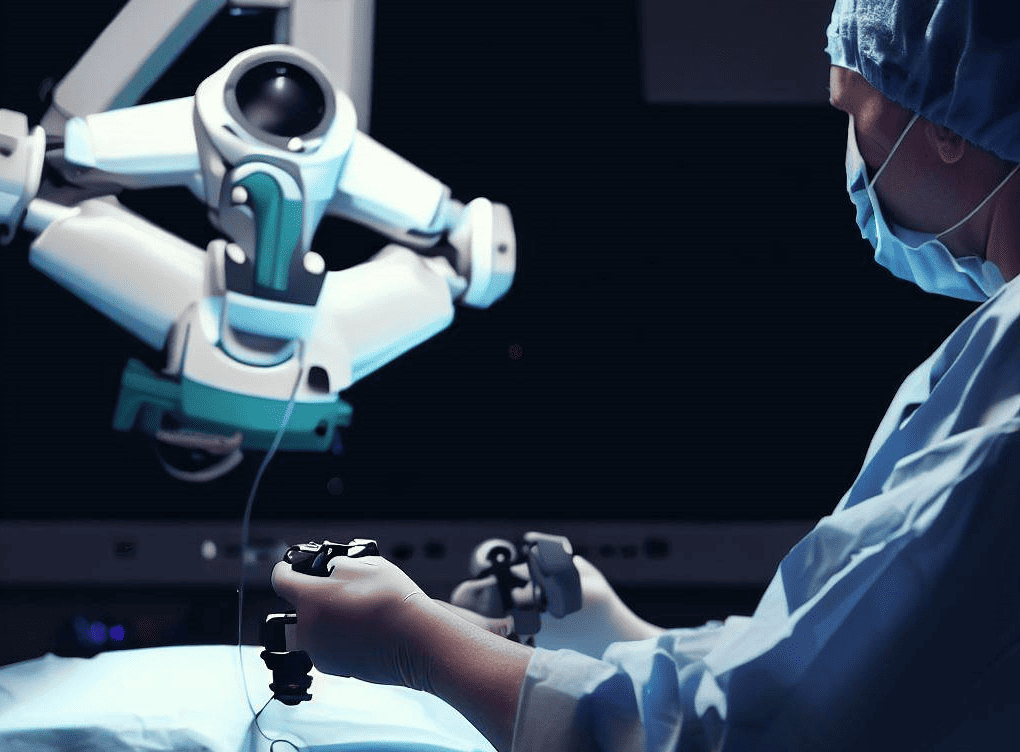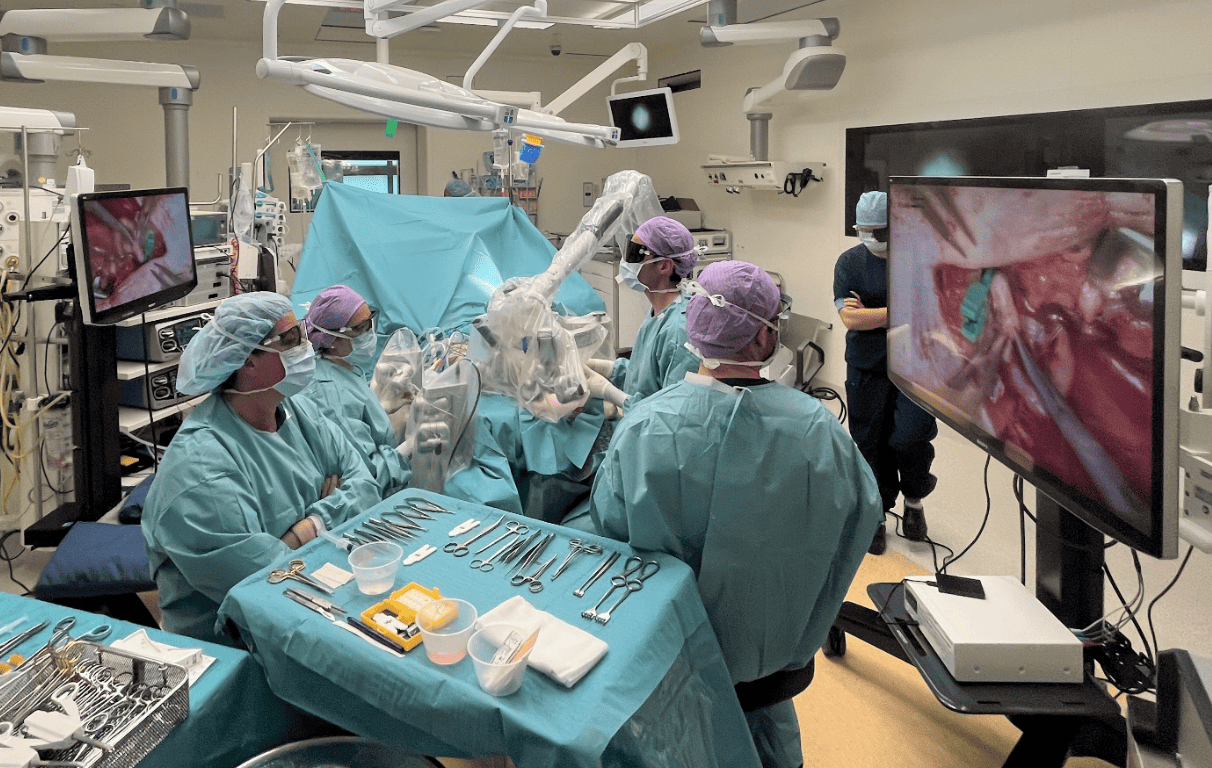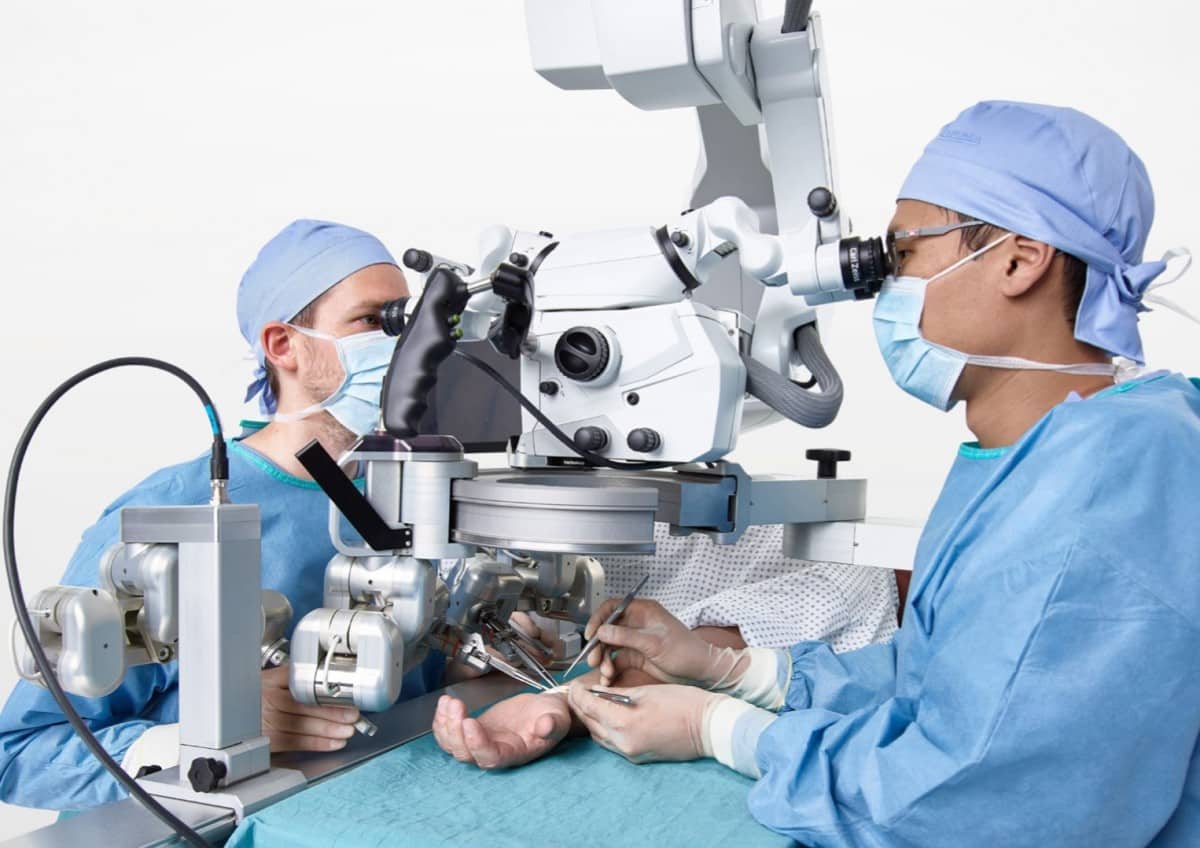
Just like their ‘neighbours’ at Preceyes, Microsure’s surgical robot MUSA has received a CE-marking, which makes it commercially available in Europe. Microsure says MUSA is the world’s first surgical robot for open microsurgery. It is developed by a team of microsurgeons and engineers, creating a high-precision robotic assistant that is compatible with current operating techniques, workflow, instruments and other OR-equipment. It enhances surgical performance by stabilizing and scaling down the surgeon’s movements during complex microsurgical procedures on a sub-millimetre scale.
“After proving safe and effective use of the device, our team is proud to announce that MUSA has received its CE mark and can be introduced to clinicians and patients in Europe”, says Dr. Ir. Raimondo Cau, founder and CTO of Microsure.
Microsure’s MUSA enables lymphatic surgery on lymph vessels smaller than 0.3 mm in diameter. Plastic surgeons at Maastricht University Medical Center+ were the first to use MUSA to surgically treat lymphedema in a patient. This world’s premier super-microsurgical intervention with ‘robot hands’ took place in 2017. Maastricht University Medical Center+ is now conducting several follow up clinical research projects with MUSA, in different fields of microsurgery. Other European hospitals are expected to start clinical studies this year.
“Microsure’s robot allows us to operate on minuscule lymph vessels and blood vessels with more ease”, says Shan Shan Qiu Shao, Plastic surgeon at Maastricht University Medical Center, “while getting better results for these complex and fatiguing interventions. Besides, it is very convenient that, within microsurgery, we can operate on vessels of every size with this robot. Most importantly, of course, this is good news for the patients concerned.”
Surgical procedures that maximally benefit from Microsure’s MUSA are complex surgeries on small tissue structures, for instance, LVA (Lymphovenous Anastomosis) surgery, pediatric vascular surgery, free flap surgery, finger and hand replantation – when human precision is the limiting factor because of physiological tremor, poor accessibility or fatigue. Various studies and pilots have proven safe and effective clinical use of the device.