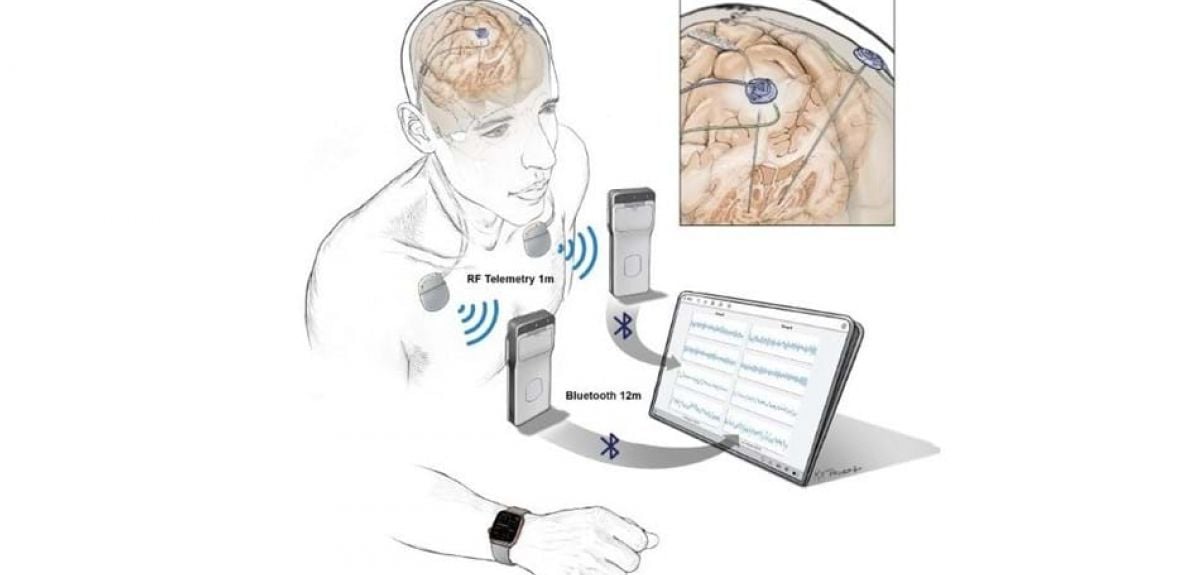
With the rise of smart watches and fitness applications, tracking the daily activity of our bodies has become quite common. Relatively less attention is given to tracking brain activity. Portable devices are now being developed to do just that. They offer new ways to treat neurological disorders, such as Parkinson’s.
The project is being spearheaded by the newly created ‘Open Mind’ consortium. This is made up of researchers from the University of Oxford, University of California San Francisco (UCSF), Brown University, and the Mayo Clinic. It recently received 3.6 million euros from the NIH Brain Initiative.
One of the challenges in treating Parkinson’s (and other neurological disorders) is the limited amount of data available. This is because the brain activity of patients is typically only measured while in a clinic. These neurostimulation devices will be developed to track brain activity of patients as they go about their daily lives. The data is then collected and can be analysed by physicians who can correlate the data with symptoms. By doing so, researchers hope to better understand the underlying issues in brain disorders and give more personalized treatment.
“Part of the reason the device is so flexible and tailorable is that the investigators write the software that controls it, which is something only device companies have been doing in the past,” says Dr Philip Starr, principal investigator and neurosurgeon at UCSF.
Intelligent therapy
One of the reasons this therapy is so promising is that it uses existing technology. Portable stimulation devices already work by providing small electrical currents to the brain. The stimulation can disrupt abnormal brain circuits that lead to symptoms. These new devices will build upon this technology but with the added feature of recording brain signals and transmitting them into data. It is useful for disorders like Parkinson’s and epilepsy but can also treat issues like chronic depression.
Safety
One hurdle with the new devices is, ironically, in their ability to be customized. It puts the complex task of customizing software and technology into the hands of researchers, who do not necessarily have the specific knowledge required. The consortium was formed, in part, to tackle this issue. It will ensure it has the proper funding and expertise to pass US Food and Drug Association (FDA) and Medicines and Healthcare products Regulatory Agency (MHRA) standards.
The idea is to work with device manufacturers and experts to also hopes to create tools and standards that can be used by the wider research community.
Also read: EmoSys: wearable technology to treat depression – Innovation Origins








