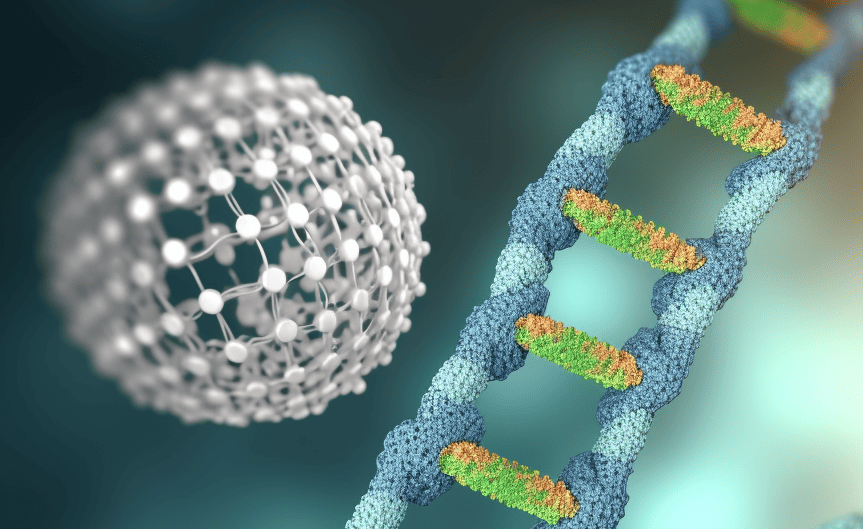
To transport drugs properly in the body, you need good packaging. The protein coatings of viruses can do that, but the genetic properties of the virus mainly determine their shape. That’s why researchers developed techniques to control the shape and size of these nanopackages so that they get the desired shape. A kind of origami with DNA, in other words.
- Researchers used “DNA origami” to create nanoscale structures that serve as molds for virus proteins.
- This allowed them to reprogram virus protein shells into different shapes like tubes and donuts.
- Cryogenic electron microscopy enabled precise visualization of the nanostructures down to individual molecules.
The high-tech tinkering was carried out jointly at Aalto University (Finland) with researchers from the University of Helsinki (Finland), Griffith University (Australia), Tampere University (Finland), and the University of Twente (Netherlands). The researchers managed to reprogram the protein shells of plant viruses into different forms. They folded DNA at the nanoscale to serve as a mold around which the protein sheaths formed. They published their results in the scientific journal Nature Nanotechnology.
DNA Origami
The researchers used “DNA origami” structures. These structures are only tens to hundreds of nanometers in size and are made entirely of DNA. By folding the DNA precisely into the desired shape, you create a template to which virus proteins can attach. “It’s like real origami in which you use flat paper to make fascinating structures in 3D. Only here, we do it with solid DNA,” explained Jeroen Cornelissen of UTwente.
The virus proteins ultimately became much more flexible than previously thought. “We managed to make different structures with a protein shell: straight tubes, but also a donut, for example. The latter is an almost opposite shape from the spherical structure that the protein mantle normally has,” Cornelissen said. It’s a simple but very effective strategy to give the virus proteins different shapes.
Cryogenic electron microscopy
Using cryogenic electron microscopy, the researchers visualized the formation of the nanostructures very precisely, down to the level of individual molecules. At around -200 degrees Celsius, they managed to measure the smallest modifications. This is the first time highly ordered proteins have been imaged this way.
The researchers see great potential in the technique. “Our approach is flexible and not limited to one type of protein, as we show with proteins from four different viruses,” adds Aalto professor Mauri Kostiainen. “Moreover, we can adapt the templates for more different applications, for example, by integrating RNA into the origami. Useful or site-specific proteins can attach to the RNA for even more complex shapes and properties.”