The human body is made up of billions of cells. Whose genomes can, in the worst-case scenario, change pathologically in such a way that they divide uncontrollably. This division occurs in cancer diseases, for example. But also in viral infections. The cause of the disease is found in the affected cells. Medication is then administered to try to destroy these cells.
However, there is hardly a drug that does not have one or more adverse side effects. Just think of chemotherapy, whose side effects usually include complete hair loss. One reason for these often-unwanted side effects is that the drugs do not only attack the diseased cells that they are supposed to destroy. The therapy affects the entire body and healthy cells are also damaged.
Stable nano-packaging for medication
A research team led by Prof. Oliver Lieleg, Professor of Biomechanics and a member of the Munich School of BioEngineering at the Technical University of Munich (TUM), and Prof. Thomas Crouzier of the KTH Royal Institute of Technology in Stockholm, have now succeeded in developing a stable nano-packaging for drugs. This allows the active ingredient to be released only within the affected cells. “The drug carriers are accepted by all the cells,” explains Lieleg. “But only the diseased cells should be able to trigger the release of the active agent.”
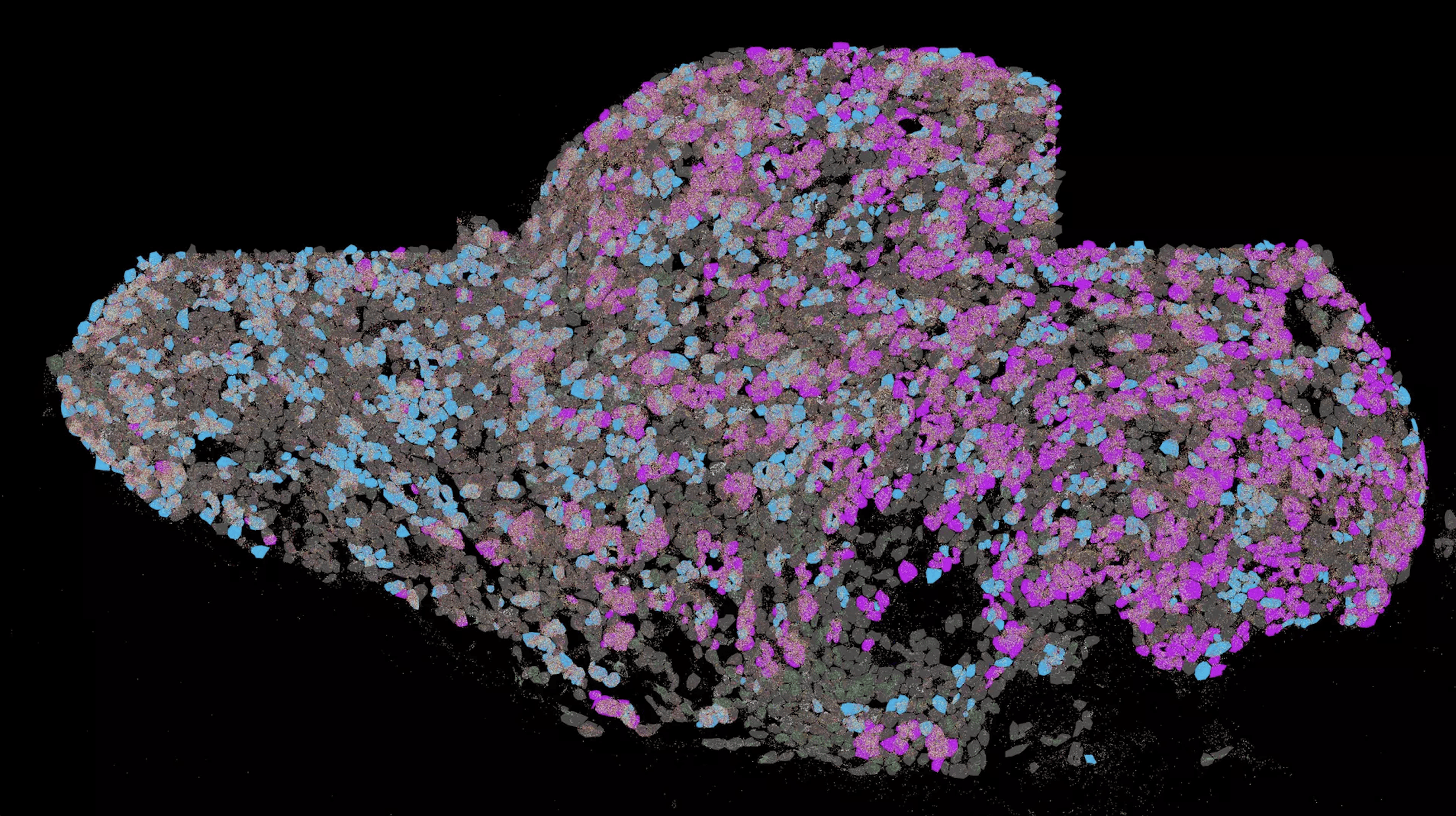
This mechanism has already worked in a trial that used tumor model systems made from cell cultures. To do this, the scientists first packaged the active ingredients using so-called mucins. These are made up of a protein spine-like structure that sugar molecules attach themslleves to. Mucins are the main components of the mucus that forms in the mouth, stomach, or intestine, for instance. “Since mucins occur naturally in the body, opened mucin particles can later be broken down by the cells,” Lieleg explains.
Another important component is also found in the body: deoxyribonucleic acid (DNA). It contains our genetic code. For the ‘active ingredient packaging’, the researchers synthesized DNA structures with the required properties and attached them chemically to the mucins. “If glycerol is now added to the solution containing the mucin DNA molecules and the active ingredient, the solubility of the mucins decreases, they fold up and enclose the active agent.” The scientists continue: “The DNA strands bond to one another and thus stabilize the structure so that the mucins can no longer unfold themselves.”
Lock and key
Only the right ‘key’ can reopen the DNA-stabilized particles. It is only then that the encapsulated active ingredient molecules can be released. Here, what are known as microRNA molecules serve as the key. That is because RNA (ribonucleic acid) is very similar in structure to DNA and plays a major role in protein synthesis in the body. However, it is also able to regulate other cellular processes.
“Cancer cells contain microRNA strands whose structure we know precisely,” explains Ceren Kimna, lead author of the study. “In order to use them as keys, we modified the lock accordingly by meticulously designing the synthetic DNA strands which stabilize our medication carrier particles.” The DNA strands are designed in such a way that the microRNA molecules can bind to them, thereby breaking the existing bonds that stabilize the structure. The synthetic DNA strands in the particles can also be adapted to microRNA structures that occur in other diseases, such as diabetes or hepatitis.
Further studies are needed
However, it will be some time before this new method can be used on patients. Before this can be done, the researchers emphasize that further studies in the laboratory with more complex tumor model systems are needed.They also want to explore “further modifying this mechanism to release active agents in order to improve existing cancer therapies.”