For small injuries such as cuts or abrasions, there are plasters that are put on the wound. The rest is done by the body’s own self-healing powers. However, it becomes somewhat more difficult with larger wounds, for example when nerve tracts are severed. Then slightly more complex methods are necessary, such as sewing the nerve stumps together. Scientists at the Max Planck Institute for Polymer Research and the Institute of Physiological Chemistry at the University of Ulm have now developed a novel biomaterial that could support the body’s self-healing powers.
Accidents often lead to injuries of the so-called peripheral nervous system, meaning nerve tracts are partially or completely severed. In such cases, the chances of recovery strongly depend on the severity of the injuries: Are the nerve tracts still partially connected? How large is the gap between the two nerve endings? Nowadays, if the distance is between a few millimetres and centimetres, surgery is generally performed. The nerve ends are sewn together again to bring them as close together as possible. Then the body’s own forces can close the remaining small gap by forming cells. This promises at least partial regeneration.
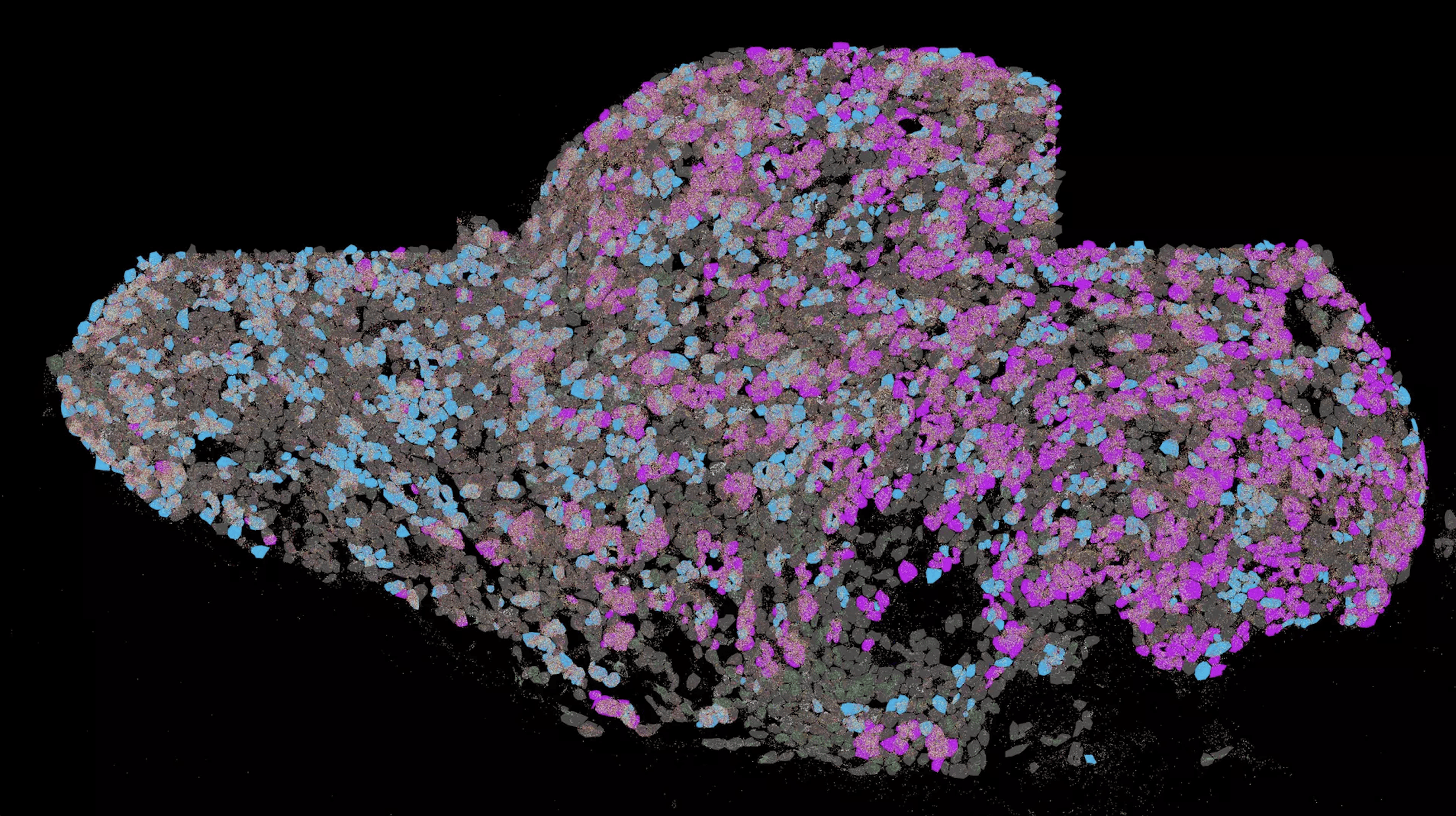
Now researchers are working on developing fluids that contain so-called nanofibers to additionally support wound healing. Nanofibers are molecular strands dissolved in water and consist of so-called peptides, which are short chains of amino acids as they also occur in human proteins. According to the scientists, these chains “serve as a scaffold or adhesion surface for cells and can form a two-dimensional lattice or three-dimensional network to which nerve or muscle cells can adhere”. This developed fluid is non-toxic to the human body and can be injected into wounds where it remains for many weeks before being degraded by the body’s own processes.

MANY POSSIBLE COMBINATIONS
The question the researchers had to solve in order to create a peptide-based bionic network was to find the right combination of molecules. From the multitude of possible combinations – so-called sequences – they had to identify those that offered both good biocompatibility and optimal cell adhesion.
The scientists explained that they first produced a series of nanofibers with systematic changes in their peptide sequence and tested them in cell cultures. “Using detailed molecular analyses and a computer-assisted algorithm, it was possible to identify recurring features in the molecular structure that are highly suitable for the regeneration of nerve cells. Subsequently, they examined the identified peptide sequences in cell tests for their ability to support neuronal growth.
“Our bionetwork can be imagined as a trellis for tomato plants,” explains group leader Christopher Synatschke. “Without grids, the plants cannot grow upwards. We have selected a grid – transferred to tomato plants – to which the plant can adhere particularly well. On a miniaturised scale, our material helps the nerve cells to bridge the gap between two nerve endings.”
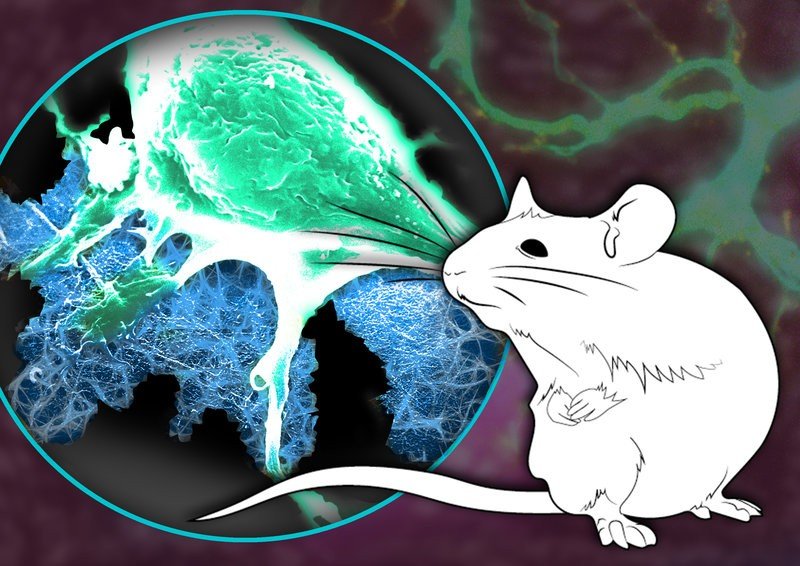
PRACTICAL TEST ON MICE
In subsequent practical tests, in which the functionality of the best material was tested, one facial nerve of mice which controls the muscle responsible for the whiskers was severed – during a “minimal surgical intervention,” as the scientists emphasize. Over the next few weeks, mice injected with biomaterial into the artificially created space between the nerve endings showed a faster and more comprehensive healing process than untreated mice.
The researchers suspect that the peptide chains produced lead to the body’s own growth-promoting proteins remaining longer in the wound. “In the future, it would therefore be conceivable to functionalise the chains in such a way that, in addition to the scaffold structure, cell growth-promoting molecules can also be introduced into the biomaterial in order to further increase its healing potential,” said Synatschke and his colleagues. In addition, they hope to be able to develop a method that can also be transferred to human nerve damage.