For the most part, the treatment of diseases still follows a standard procedure, whether they are chronic ailments or acute complaints. To a large extent, this is also associated with high costs, both for long hospital stays and for medications. Not to mention their side effects, which can vary considerably from patient to patient. For the treatment of diseases such as rheumatoid arthritis, chronic headaches, asthma or even Parkinson’s disease, doctors have therefore been increasingly resorting to neuromodulation, i.e., the stimulation of nerves by electrical impulses.
This personalized treatment method not only reduces hospital stays and healthcare costs, it also brings important benefits for patients. In addition to special drugs, this is primarily done using so-called electroceuticals, i.e., microimplants that use electricity to provide personalized, localized treatment without triggering any side effects in the body. Fraunhofer IZM researchers are now developing a new generation of microimplants that rely on ultrasound instead of electricity as part of the Europe-wide, EU-funded Moore4Medical project. As a result of this new technology, these implants can also be charged completely wirelessly.
Implants not just close to the skin
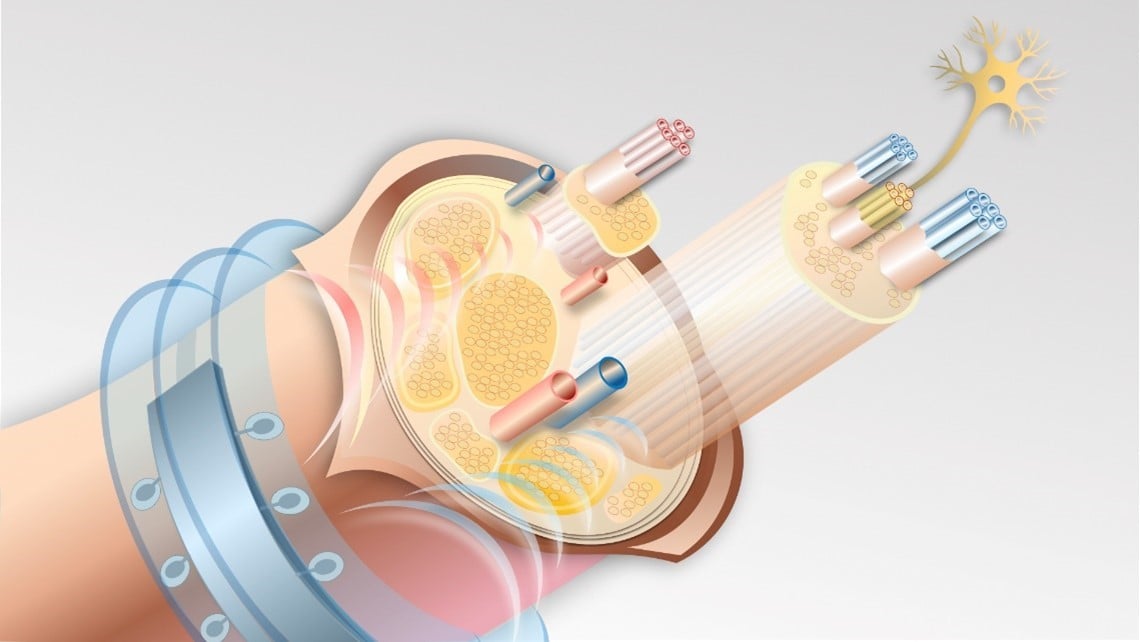
Ultrasonic waves penetrate the body from the outside, which means they also reach the microimplant. Unlike conventional battery-charged devices, such implants can therefore also be charged from the outside. This eliminates the need for battery replacement surgeries or charging with a cable. In addition, since no additional battery is needed, these microimplants can also be designed much smaller. This means they can precisely stimulate tiny nerves even as small as 20 microns. Another advantage is that, unlike battery-powered solutions, the implants can also be inserted deep inside the body. Conventional implants can only be inserted close to the skin.
There is also a slight difference in the way they work. While battery-powered implants emit direct pulses of electricity, these new implants vibrate with high-frequency sound, with this motion then being converted into electrical energy for the microimplant. According to the scientists, one challenge is to optimally align the vibrating microstructures to avoid high losses during energy transfer. Another hurdle is that the overall size of the implant must not exceed a few millimeters. This means that ultrasound transducers, electrodes for recording neuronal activity and all other components must be miniaturized to a few millimeters, integrated and built to last.
Further developing basic components for numerous applications
Researchers are currently working on assembling the right materials for the prototype. These must be biocompatible while also being suitable for encapsulation and energy transfer through sound waves. Next, several transducers must also be built in groups “so that a combination of the electronic components and thus a more concentrated emission of the ultrasonic wave is achieved.”
The project will run until June 2023, by which time it is hoped to create a toolbox of sorts that will enable faster, cheaper and more powerful medical technology. In the future, the basic building blocks developed as part of Moore4Medical could be further developed in other research, the scientists believe. For example, for specialized applications in wireless microimplants, organ-on-chip, 3D ultrasound, sustained monitoring using sensors, drug adherence through intelligent delivery and also X-ray-free surgery with optical sensing.
Also of interest:
Cancer research – personalized therapies possible (German only)
Faster detection of heart attack with personalized algorithm
Digital twin for personalized therapies (German only)