
Green hydrogen is a serious contender to become a renewable energy carrier. For example, we could use it to store solar and wind energy during peaks in order to utilize the energy when the sun is no longer shining and the wind is calm. This would require more electrolyzers that can efficiently and flexibly handle fluctuations in power supply.
However, the electrolyzers that can already do so use rare and expensive raw materials for their electrodes, such as platinum and iridium, while more affordable alkaline electrolyzers struggle to operate flexibly. This is where a team of TU/e researchers, led by Thijs de Groot (Chemical Engineering & Chemistry), may have now found a solution.
De Groot: “You can make alkaline electrolyzers with relatively cheap raw materials, which makes them very suitable as a sustainable choice. That’s exactly why I wanted to do research on them – to see if we can make them suitable for flexible energy storage.”
- Green hydrogen requires efficient electrolyzers to handle fluctuations in power supply;
- Current affordable alkaline electrolyzers find it difficult to handle electricity supply flexibility and use expensive materials for their electrodes;
- TU/e researchers have found a solution by optimizing the distance between the membrane and the cathode (“gap”).
The challenge: the hydrogen leak
So, what’s the most difficult thing about these alkaline electrolyzers? To answer this, we have to go back to the basics of what happens in an electrolyzer, where a cathode and an anode are placed in a salt solution with a membrane between them. If you apply enough voltage, a current flows; hydrogen (H2) forms at the cathode and oxygen (O2) at the anode.
The membrane is supposed to keep the hydrogen and oxygen from coming together. However, no membrane is able to keep oxygen and hydrogen perfectly apart, which can potentially create dangerous situations.
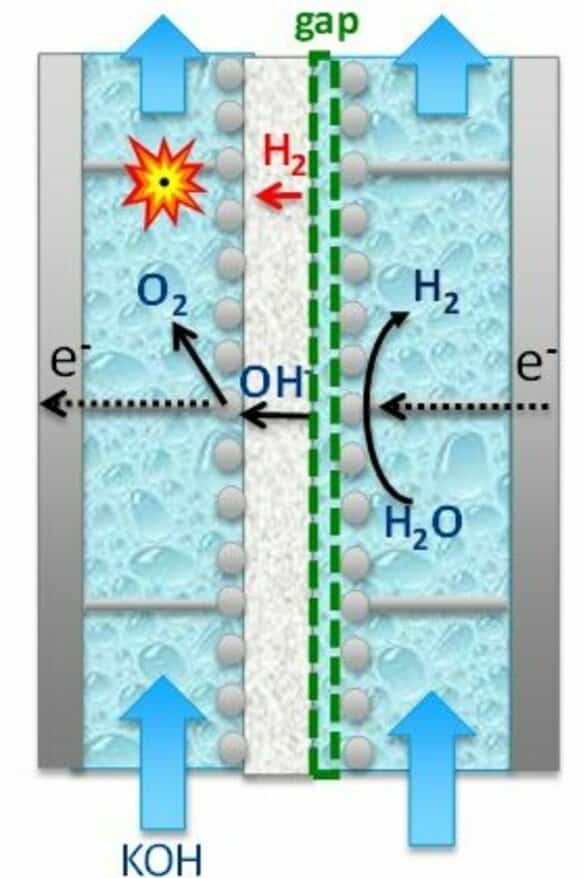
The basic principle of an electrolyzer
By applying a voltage to the electrodes (with the anode on the left and the cathode on the right), negative ions from the electrolyte solution move to the cathode and positive ions move to the anode. At the cathode, a chemical reaction takes place in which hydrogen is formed. At the anode, the reaction results in oxygen.
Explosive mixture
The danger lies in the hydrogen leaking through the membrane and ending up on the oxygen side (known as hydrogen crossover). If the amount of hydrogen in the oxygen exceeds four percent, an explosive mixture is created.
Hydrogen leakage always takes place to some extent but is especially dangerous when the electrolyzer is not running at full power. In that case, less oxygen is made, so the hydrogen leaking through the membrane is not diluted as strongly. The concentration then approaches the explosive limit more quickly.
To mitigate this danger, the preference is now for alkaline electrolyzers to run at full power. They can be turned up or down a bit but cannot handle sudden large changes. This makes it more difficult to connect them directly to solar or wind farms. It is also tough to operate the electrolyzers with hydrogen at very high pressure because even more hydrogen will then leak through the membrane. But perhaps a change is coming.

The solution: managing the gap
To enhance the flexibility of alkaline electrolyzers and ensure their safe operation when connected to variable power supply, we aim to control the hydrogen crossover. The distance between the membrane and the cathode – the gap – is very important to this.
In the past, research has been done on these gaps at electrodes. Those studies focused mainly on the efficiency of electrolysis. And, this efficiency is greatest with a zero gap at the cathode.
More flexibility, no explosion hazard
However, if there is no gap, a lot of gas will move through the membrane. This is due to a high supersaturation of hydrogen near the membrane. Supersaturation is the same phenomenon that occurs when you open a bottle of beer or a carbonated soft drink. So, this supersaturation must be brought down if you want to make your electrolyzer more flexible without the danger of an explosion.
De Groot: “For this reason, Rodrigo Lira Garcia Barros – PhD student and first author of the article – systematically investigated the effect of gap size during his doctoral research. This concerned, in particular, the effect on the amount of hydrogen moving through the membrane and the performance of the electrolyzer. It then turned out that with a small but measurable gap at the cathode, we can greatly reduce the leakage of hydrogen through the membrane – and with an acceptable loss in performance! In the end, you’re left with a more functional and, above all, more flexible electrolyzer.”

Proof
De Groot: “That’s the conclusion of our scientific analysis, so it’s now time for the next step: creating a proof of concept. That’s what we’re going to do in our lab, through which we expect to be able to prove that a flexible electrolyzer is scalable to industry.” Scalable electrolyzers can also be made in different sizes. For example, very large ones could be used for central energy storage, just as power plants now operate centrally in our grid, or smaller ones could accommodate peaks in power generation at the neighborhood or wind farm level.
The next step: high-pressure electrolysis
De Groot enthusiastically goes on to talk about a new European project that is also starting at TU/e: high-pressure electrolysis. “The hydrogen released in electrolysis is a gas. But gas can only be used, stored, and transported efficiently if it is stored at a sufficiently high pressure.”
“We currently need a compressor for that, in addition to the electrolyzer itself. Those devices are expensive, noisy, and unreliable. So, what if we could make an electrolyzer that immediately delivers the hydrogen at a high pressure? A few years back, I thought that this wouldn’t be possible, just as I thought that we wouldn’t be able to make a super-flexible alkaline electrolyzer. We managed to do the latter, so I’m very much looking forward to again working with students on this new challenge.”
The original version of this article was published on the TU/e website.









