In an update on developments around a potential vaccine for COVID-19 based on VSV, New York-based Humane Genomics is reporting a new success in the lab. By showing that the proposed vaccine is binding the ACE2 receptor, the lab has proven that the vaccine has the typical COVID-19-like spike protein on the outside. “This is super exciting”, CEO Peter Weijmarshausen says. “Because if the theory holds, that our immune system can be trained by a weak virus with the spike protein, then we are in good shape.”
Dutchman Peter Weijmarshausen is the former CEO and founder of Shapeways, an Eindhoven and New York-based company for 3D printing concepts. He co-founded Humane Genomics last year, in an effort to make synthetic, artificial, viruses. The first application would be as cancer therapies, but recently the company shifted to COVID-19 as well. Read more about Weijmarshausen and Humane Genomics here.
In an explanation of the process, Weijmarshausen says the physical outside of the virus SARS-CoV-2 is characterized by the spike protein. “It is the spike protein that enables the virus to infect cells. How does it do that? The spike protein matches with the so-called ACE2 receptor on cells. You could say that the ACE2 receptors are the lock and the spike protein is the key. Cells that have the ACE2 receptor can be infected by a virus with the spike protein.” Human lung cells, for example, have ACE2 receptors.
“It is our belief that a successful vaccine should have this spike protein, but of course, should not be pathological. The immune system is triggered by the outside of the virus and it will be trained to recognize it and kill it. This idea is the basis of our vaccine. Putting the spike protein on the outside of VSV (a non-pathological virus) enables you to train the immune system without side effects. By putting our vaccine virus together with cells that have the ACE2 receptor we can see if they can infect them. And that is exactly what we have shown in the lab.”
This Humane Genomics video shows what happens:
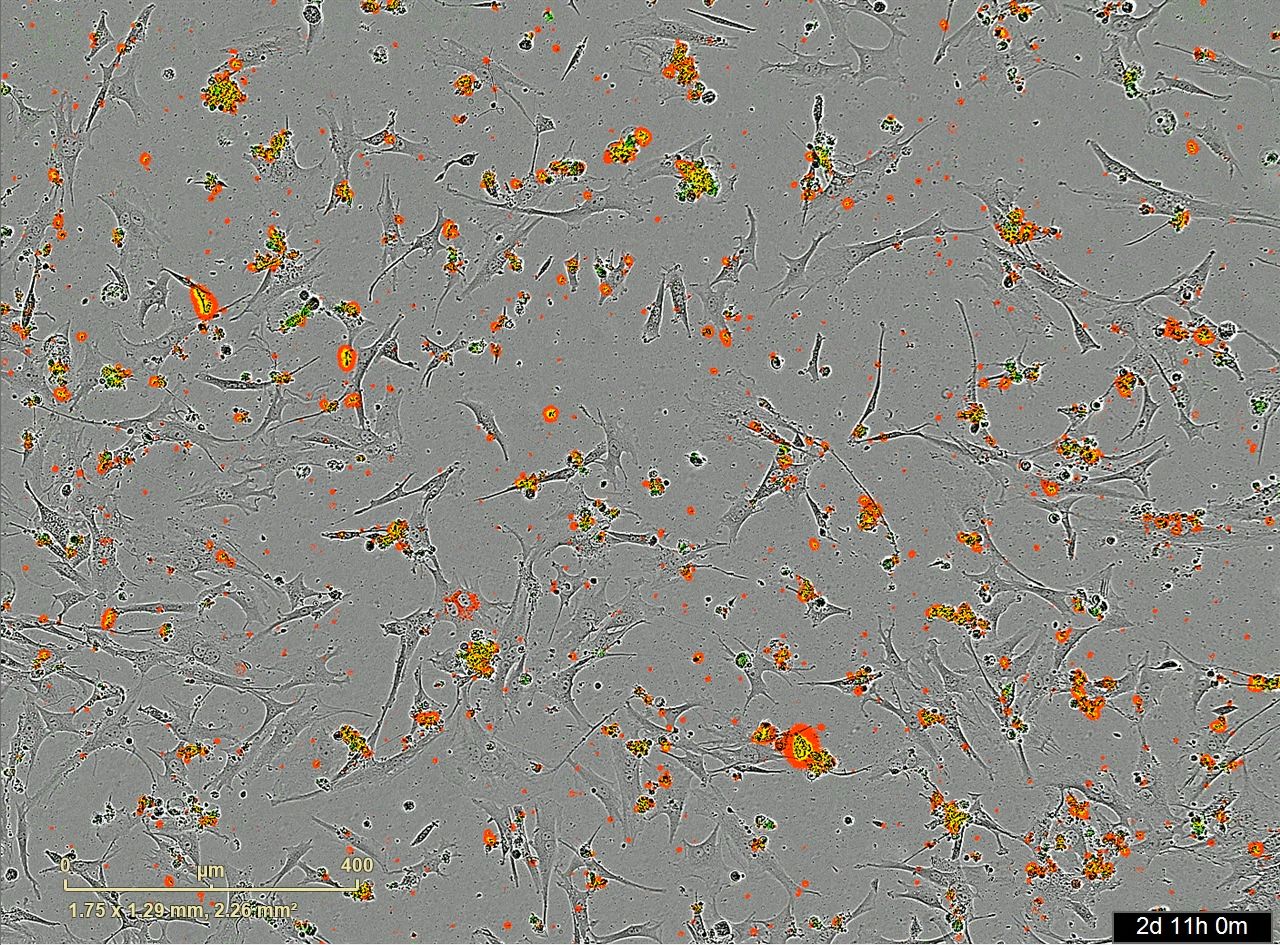
Red is the ACE2 receptor on the cells. Green is the Spike-S protein on the vaccine. The yellow patches are co-localization of both cells with the ACE2 receptors and the vaccine.
The next step for Humane Genomics is to conduct safety testing in animals (lab rats), followed by testing in guinea pigs to see if the vaccine provides immunity and protection. “If all goes well, we hope to start the first phase of clinical trials.” No specific date is mentioned.
In their update, Humane Genomics says the company is still looking for help, like partnerships with clinical researchers to host clinical trials, and companies who can make small batches of the virus at GMP grade, with the potential for scaling to larger quantities.








