Antibodies play a decisive role in our lives and our health. After suffering from a viral infection, the body usually builds up these antibodies of its own accord in order to protect itself from further infection. The same principle also applies to vaccinations. They are designed to stimulate the development of antibodies without the body having to endure the respective disease. This is why these proteins play a pivotal role. Especially in medicine – as is currently being highlighted in the worldwide search for a vaccine against COVID-19.
These so-called immunoglobulins can be used in many ways to combat pathogens because they are able to detect and tag foreign substances. This enables our immune cells to recognize and fight these substances over the long term. At the same time, the immunoglobulins also help to destroy potential invaders with the help of other immune cells. The antibodies are produced in dedicated production units, in what are known as plasma cells. They are produced at the rate of several thousand per second.
GN1 as an agent in the immune system
A research team led by Dr. Josef Penninger of the Institute of Molecular Biotechnology at the Austrian Academy of Sciences (IMBA) has now focused on the role of a specific protein in the production of antibodies: the JAGN1 gene. Penninger was previously able to identify this protein as an agent in the body’s immune system of patients with Severe Congenital Neutropenia (SCN). He did this in collaboration with researchers headed by Christoph Klein at the Dr. von Haunersches Children’s Hospital in Munich, Germany. SCN is caused by a mutation in the JAGN1 gene. This leads to abnormally low white blood cell levels in affected patients. Because of these neutrophils, they end up with severe infections as their immune system cannot defend itself adequately against bacterial and fungal infections.
In the current study, the researchers have examined the role of JAGN1 in relation to so-called B cells. These are white blood cells that can transform into plasma cells once they detect foreign substances (antigens). Examples of antigens include chemicals, bacteria, viruses, and pollen. Each of the plasma cells can then produce thousands of specific antibodies per second that target a particular antigen.
This mass production takes place at a specific location in the cell, the endoplasmic reticulum. The resulting antibodies are then lined with sugar molecules on their Fc region. This is a region that interacts with other parts of the immune system. This process, known as glycosylation, makes it easier for the antibody to bind to other immune cells. Which in turn amplifies the body’s defense response. Specific sugar structures that are attached to proteins are known to influence protein stability. Communication between cells and their environment is also affected.
Fundamental understanding of antibodies due to rare genetic defects
“When we knocked out JAGN1 in B-cells of mice, we were able to measure a drastic reduction in the number of antibodies,” Senior Research Associate at IMBA Dr. Astrid Hagelkruys goes on to explain. She is the first author of the study of the publication, which was published in the Journal of Experimental Medicine. “In addition, their glycosylation pattern – the addition of specific sugar molecules – was altered on antibodies of JAGN1-deficient B-cells.”
“JAGN1 appears to affect the antibody factories in the cells,” Dr. Penninger says. “To our surprise, this change in the sugar structure also leads to a better ability of the antibodies to bind to other immune cells and strengthens the defense response.”
The scientists were also able to confirm the same mechanism in human subjects. “Rare genetic defects occur in only a handful of people. But sometimes fundamental principles of biology can be derived from them,” says Penninger. “But they can sometimes help us decipher basic principles of biology. In this case, we were able to prove that a certain gene affects the endoplasmic reticulum and is therefore essential for the mass production of antibodies. ” At the same time, the researchers learned that the “sugar coat” of antibodies is also altered: “Which has an important effect on how such antibodies actually work in the body.”.
Original Publication: ‘A crucial role for Jagunal homolog 1 in humoral immunity and antibody glycosylation in mice and humans.’ Hagelkruys et al, Journal of Experimental Medicine, 2020. DOI: 10.1084/jem.20200559
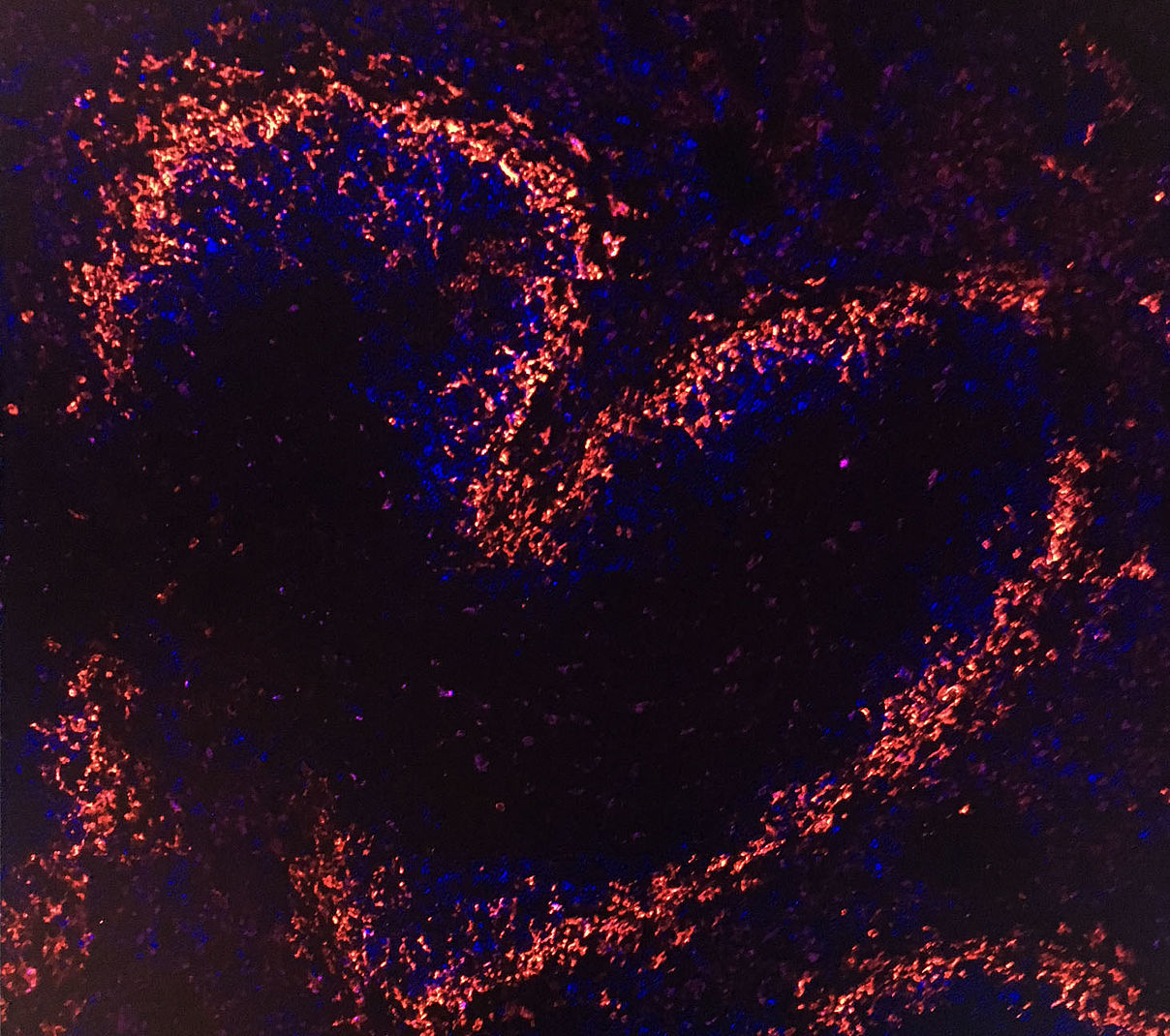
Title image: A tile scan of a mouse spleen stained with MOMA1 (red) and CD23 (blue).