No sustainable energy without electrocatalysts: For the production of green hydrogen, catalysts are needed to control the process of splitting water into oxygen and hydrogen. However, the structure of the catalyst may change when an electrical voltage is applied, which also has a decisive influence on the catalytic activity. This phenomenon is poorly understood – so far. A research team led by the University of Duisburg-Essen and the University of Twente has investigated the crystal facet-dependent surface transformation and how this controls the activity of the oxygen evolution reaction. Their results have now been published in Nature Communications.
- A research team studied how the surface transformation of catalysts affects the efficiency of splitting water into hydrogen and oxygen, which is crucial for green hydrogen production.
- The study focused on perovskite-class catalysts, finding that their surface transformation during the reaction impacts oxygen evolution reaction activity.
- This research is key for developing more efficient and sustainable catalysts for green hydrogen production.
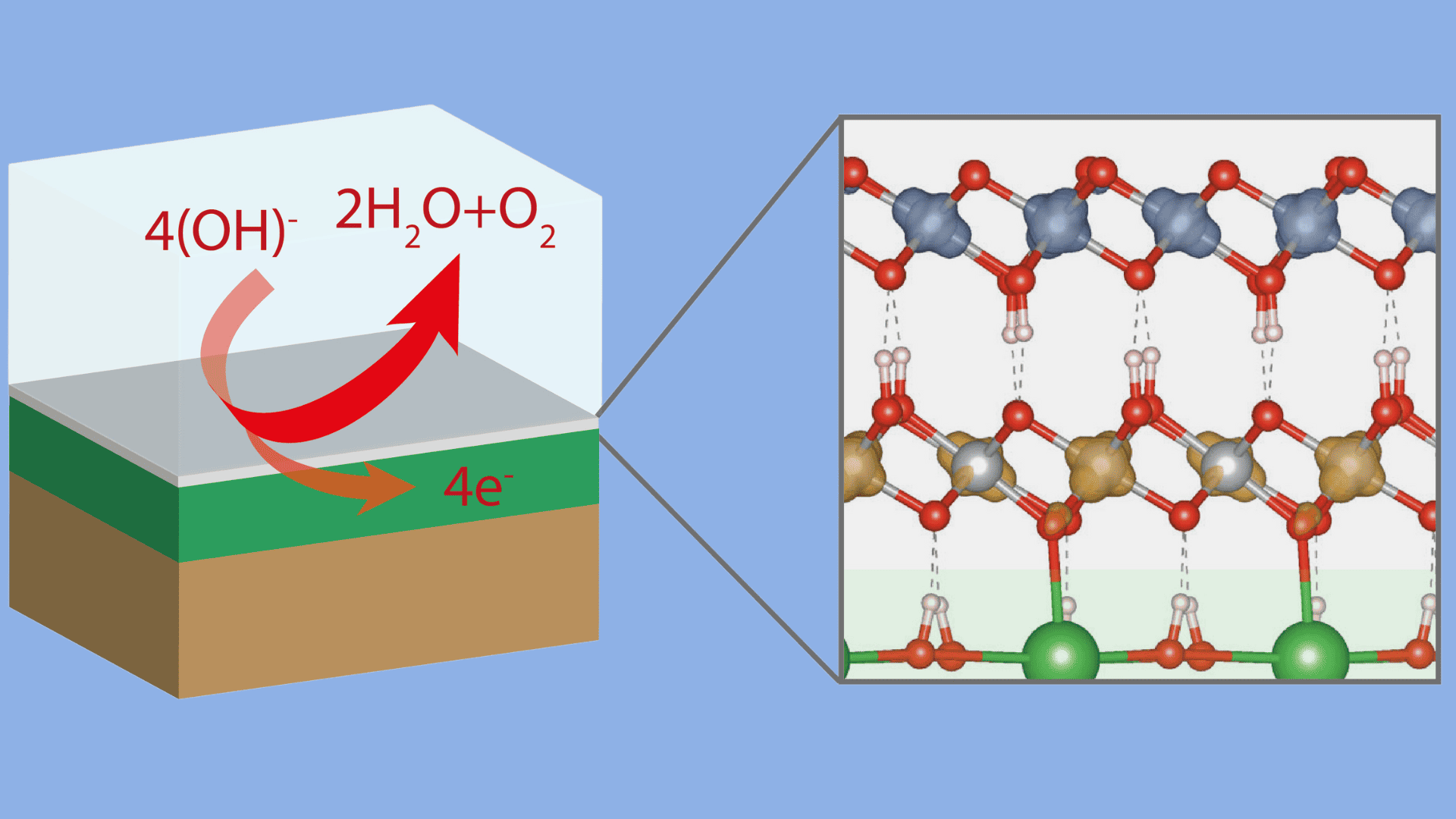
The goal is to optimize catalysts in a targeted manner by producing nanoparticles with a specific surface orientation – thus producing cost-effective and sustainable hydrogen. Researchers from UDE, University of Twente, Forschungszentrum Jülich, and Helmholtz Zentrum Berlin für Materialien und Energie have taken a major step towards achieving this goal. They investigated the complementary half-reaction of the production of green hydrogen – the formation of oxygen. This partial reaction is more complex and requires highly efficient, cost-effective, and environmentally friendly anode materials. However, the structure of the original catalyst often changes during this process – a phenomenon that is still poorly understood.
The scientists proved that this transformation depends on the orientation of the catalyst’s surface and impacts the oxygen evolution reaction (OER). Higher OER activity means that the catalyst can generate oxygen more effectively, improving the efficiency and sustainability of the hydrogen production process.
Perovskite
In their study, the scientists used lanthanum nickel oxide films (LaNiO3) – a crystalline material of the “perovskite” class used in electrocatalysis. The researchers discovered that the uppermost layers transform during the reaction and exhibit different OER activity. The octahedron-shaped building blocks connect via the edges instead of the corners. This oxyhydroxide structure was modeled using quantum mechanical simulations on various perovskite surfaces by Achim Füngerlings in the working group of Prof. Dr Rossitza Pentcheva on the UDE’s high-performance computer (magnitUDE). “The results show that for the facet with the highest activity, the transformed layer fits much better on the underlying perovskite,” says Füngerlings.
“This significantly influences the electronic and magnetic properties and explains the improved OER activity,” explains Pentcheva. “The close cooperation of experiments and simulations was crucial to elucidate the underlying mechanisms and properties of the surface transformation.” The study results are important for the future optimization of catalysts by producing nanoparticles with a specific surface orientation.







