One thing certain from the day we are born that applies to everyone is that the biological process of aging begins immediately and progresses a little further with each passing day. The older we get, the more pathological changes occur. These range from heart weakness to the loss of nerve cells to reduced metabolic activity. And this process is irreversible.
One reason for these age-related changes is that certain cellular cleansing processes no longer function as well as they did in younger years. This so-called autophagy is a kind of recycling system that breaks down and recycles superfluous or damaged components of the cell. This keeps the cells fit and protects them from many diseases. Unfortunately, however, autophagy falters over the years with increasing age.
A research group of neurophysiologists, nuclear physicians and molecular biologists led by Professor Evgeni Ponimaskin of the Institute of Neurophysiology at the Hannover Medical School (MHH), Germany, has now found a way to revive autophagy. The body’s own substance spermidine can have a genuine anti-aging effect.
Mice with less organ damage and hair loss
Spermidine is found in every cell in the body, but its concentration decreases with age. “In our study, we investigated how prolonged spermidine administration affects organs commonly affected by aging and the process through which this occurs,” explains Professor Ponimaskin. The scientists administered spermidine to aged mice for six months through their drinking water. The result showed that these mice showed significant anti-aging effects compared to non-treated, age-matched animals. “Spermidine supplementation ensured that the animals developed less kidney and liver damage and exhibited a better performance-enhancing glucose supply in the brain,” explains the study leader. Age-related hair loss was also significantly lower than in the control group. Thanks to the spermidine supply, there were hardly any bald spots on the back, which is typical for older mice.
Protection against degradation of chromosome ends
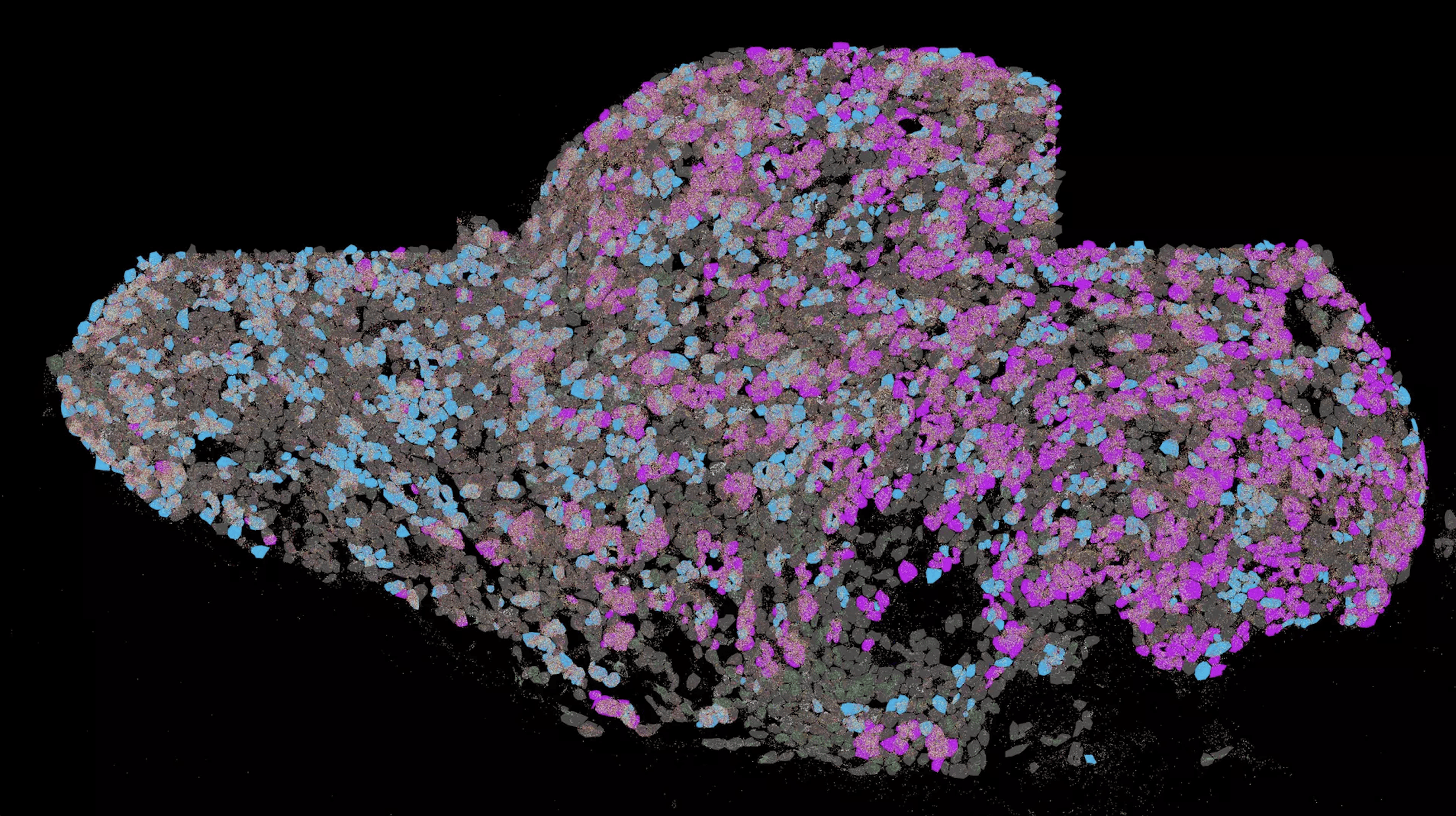
The researchers were particularly impressed by the protective effect of spermidine on the heart. They found that the cardio-protective effect of spermidine was related to reduced shortening of telomeres in heart tissue. Telomeres protect the ends of chromosomes in our body’s cells from degradation. Chromosomes are carriers of our genetic information. Each time cells divide, their ends shorten a bit. When cells stop dividing, in this case heart muscle cells, the telomeres are shortened further. At some point, they become so short that so-called programmed cell death sets in. “The telomeres in spermidine-supplemented mice were similar in length to those in young animals,” says Professor Ponimaskin. These findings could also have long-term implications for protection against age-related diseases in humans. This is because the aging processes in the cells of mice are similar to those in our body cells, so spermidine supplementation could also protect us from many age-related diseases.
What is spermidine?
Spermidine is an endogenous, natural substance first discovered in male seminal fluid, hence the name spermidine. However, it is now known to exist in all body cells and certain intestinal bacteria can also produce this substance. But the bulk must be taken in through food, for example, wheat germ, cheese, soy products and legumes. The substance activates autophagy in our cells, breaking down invading pathogens, defective proteins and cell components that are no longer functional.
The study was published in cooperation with the University of Graz, Austria, in the journal GeroScience. “Novel aspects of age-protection by spermidine supplementation are associated with preserved telomere length.”
Cover photo: Professor Evgeni Ponimaskin (left) and Dr. Alexander Wirth. The monitor shows an aged mouse without spermidine supplementation at the top and an aged mouse with spermidine supplementation at the bottom. Copyright: Karin Kaiser / MHH