In a weekly column, alternately written by Maarten Steinbuch, Mary Fiers, Carlo van de Weijer, Lucien Engelen, Tessie Hartjes and Auke Hoekstra, Innovation Origins tries to find out what the future will be like. The six columnists, occasionally supplemented by guest bloggers, are all working in their own way on solutions to the problems of our time. So that Tomorrow will be Good. This Sunday it’s Auke Hoekstra’s turn, about the power of batteries. Here are all previous episodes.
 For the first time in human history, we are no longer going to derive our energy from burning the inventory of the earth. Instead, we are going to switch to electron pumps that are directly driven by that inexhaustible source of energy high up in the air. But what can we do when the sun doesn’t shine and the wind doesn’t blow? And how do we take that solar energy on the road? Batteries offer a solution.
For the first time in human history, we are no longer going to derive our energy from burning the inventory of the earth. Instead, we are going to switch to electron pumps that are directly driven by that inexhaustible source of energy high up in the air. But what can we do when the sun doesn’t shine and the wind doesn’t blow? And how do we take that solar energy on the road? Batteries offer a solution.
Last week I tweeted about Norway’s plan to switch all domestic traffic to battery-powered electric aircraft. I think that makes perfect sense.
And this weekend, I’m working on a report focusing on switching Dutch diesel trains to battery trains: also logical. Still, I notice that many people do not yet understand that. That’s why I thought: let’s playfully clarify what’s going on with all those fantastic battery developments.
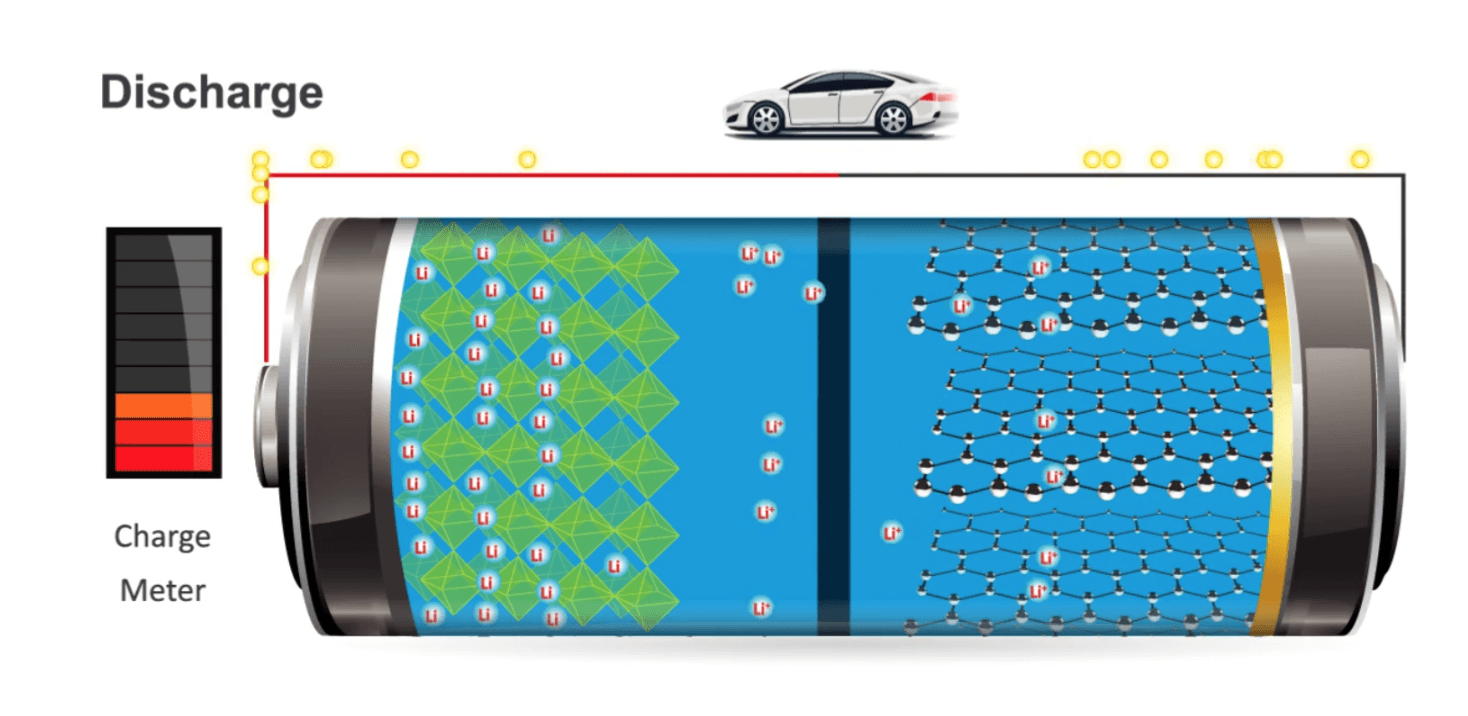
In batteries, all the work is done by so-called ions. In the case of lithium batteries, these are lithium ions. For every electron that is pumped around outside of the battery, a lithium ion swims the other way around inside the battery. For a battery of one kWh with cells operating on 3.7 volts, only 70 grams of active lithium ions are needed. So for the heaviest Tesla battery, you only need 7 kilos of lithium and you can drive 350 kilometers a couple of thousand times, after which you can recycle it! Compare that to petrol. To cover the same distance, you need 25 liters of petrol a few thousand times. And instead of recycling petrol, you have many tonnes of CO2 emissions.
Unfortunately, the 7 kilos of hard-working lithium ions also need an environment to work in, which makes the battery more than fifty times heavier than the lithium ions that perform the real work. That is why we have to talk about three things: 1) the ions at their homes, 2) their holiday destination and 3) the dangerous road in between. When you hear about breakthroughs, it’s almost always about only 1 of those 3, so it’s great if you can keep them apart.
The positive electrode: the preferred home of the ions
 The home base of the always positive lithium ions is the positive electrode shown on the left on this picture. They prefer to be there – by far. This is what we call the cathode. (Officially, this is only the cathode if the battery is discharged. When the battery is being charged, it is suddenly the anode. Very confusing. To limit the confusion, we always call the positive electrode for lithium batteries the cathode.)
The home base of the always positive lithium ions is the positive electrode shown on the left on this picture. They prefer to be there – by far. This is what we call the cathode. (Officially, this is only the cathode if the battery is discharged. When the battery is being charged, it is suddenly the anode. Very confusing. To limit the confusion, we always call the positive electrode for lithium batteries the cathode.)
We are constantly looking for cathode materials that are lighter and they often give their name to the battery. First, we had the LFP battery containing iron, but then we managed to replace it with the lighter NCM or NCA cathode. In the near future, lithium sulfur seems to be the perfect cathode because it is much lighter and cheaper, while sulfur is abundantly available. If that happens, all battery predictions about prices and weights will be turned upside down.
The anode: the hated Airbnb of the ions
But when we charge the battery, we put the house of the ions under power and they are forced to leave their house and swim to the negative electrode of the battery: their hated Airbnb called anode. We also want to make these lighter. So if you hear that we’re switching from graphite to silicon because it’s much lighter, it’s about the anode. And one day the anode may be made from the miracle material graphene. That, too, would turn everything upside down.
The electrolyte: the dangerous path the ions have to travel
The fact that batteries slowly deteriorate is partly due to the decay that all material undergoes over time, especially at higher temperatures. But it’s mainly because the journey from cathode to anode is full of dangers, as a result of which more and more ions are no longer able to finish the journey and are stranding on the way. They undertake that journey through the material that separates the cathode and anode: the electrolyte.
When you hear about problems with wear and tear and fires in batteries, it almost always has to do with the electrolyte. There is a movement towards solid-state batteries in which this electrolyte is no longer a liquid but a solid. Now it’s still a polymer (plastic) but later probably ceramic material. Once we’ve made that switch, a battery lasts much longer and will never catch fire again, even in the most extreme situations.
And to come back to that plane in Norway: with the stormy developments in the field of batteries, I am convinced that not only cars, trucks and diesel trains, but also most planes will switch to clean, quiet and sustainable electricity. The potential of batteries is truly incredible.







