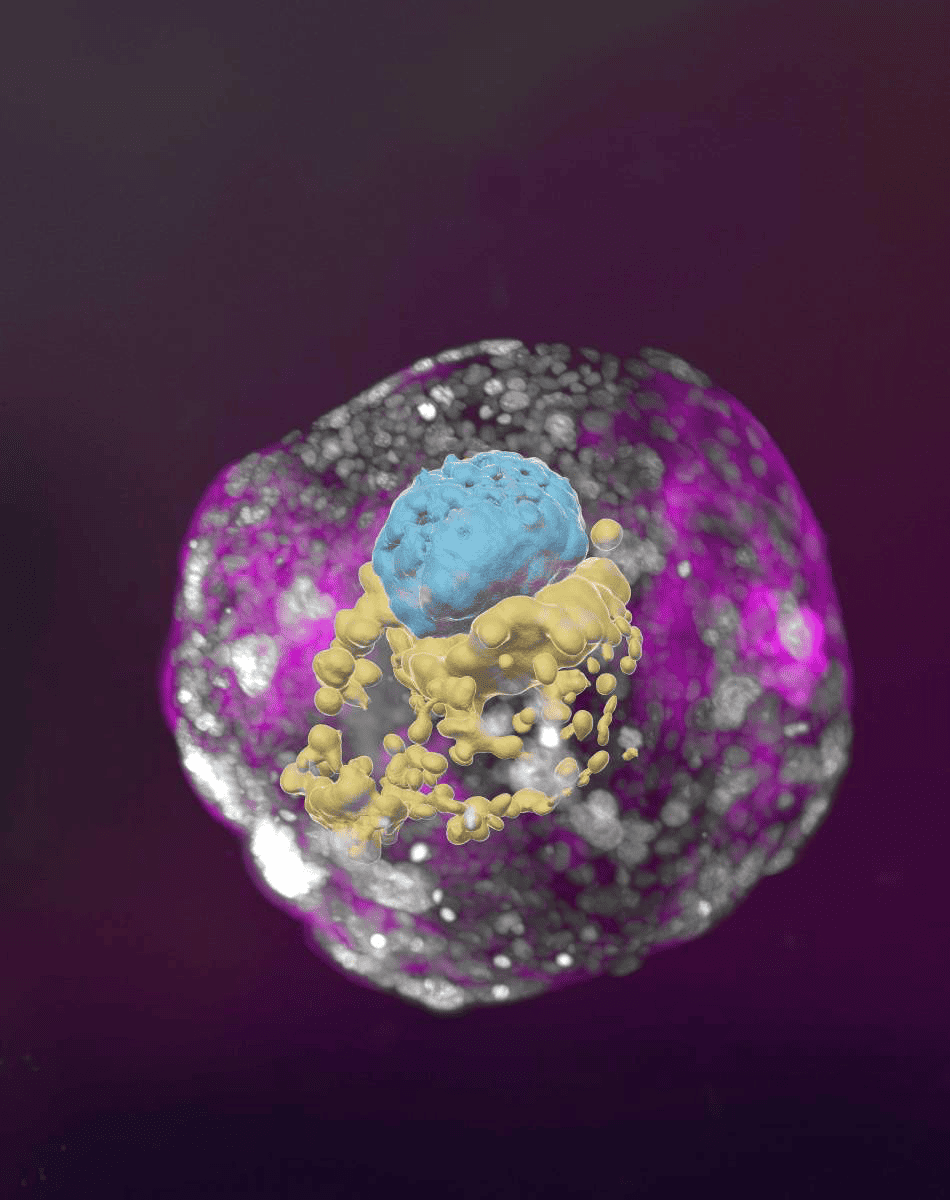
An international research team has achieved an important breakthrough within the field of stem cell research. They have obtained a type of stem cell from pig and human cells that corresponds to the very early stage of a fertilized egg cell. In doing so, these stem cells have an especially huge potential for development and could open up completely new possibilities for research.
Various cells in the body serve different purposes. However, stem cells are cells that do not yet have a specific function and can, therefore, become almost any cell that the body needs. Thus, stem cells could also play a role in the treatment of some diseases that are currently incurable. Stem cells are divided into three groups: adult, embryonic and induced pluripotent stem cells.
Adult, embryonic and induced pluripotent stem cells
Adult stem cells, also known as tissue-specific or somatic stem cells, exist throughout the body from the time an embryo is developed. These cells are in a non-specific state but are more specialized than embryonic stem cells. They remain in this state until the body needs them for a specific purpose, for example after an injury as skin or muscle cells or to “repair” organ damage. Stem cells are present in the brain, blood and blood vessels, liver, bone marrow and other areas.

Embryonic stem cells originate from the blastocyst, the embryo created four to five days after fertilization of the egg by the sperm. Before the embryo is implanted into the uterus, a mass of about 150-200 cells forms the blastocyst. This consists of two parts: the outer cell mass, which becomes part of the placenta, and the inner cell mass, which develops into the human body. The inner cell mass contains the embryonic stem cells, which have the full potential to develop into any cell inside the body. They can become blood cells, skin cells, and all other cell types that the body needs. Embryonic, so-called pluripotent, stem cells are able to develop into more various cell types than adult stem cells are able to.
Induced pluripotent stem cells (iPS) are produced in a laboratory using skin cells and other tissue-specific cells. These cells behave similarly to embryonic stem cells which means that they could be useful in the development of a number of therapies.
Breakthrough
Stem cells taken from embryos by scientists usually originate from additional embryos created by in vitro fertilization (IVF). However, these cells, taken from the blastocyst and cultured in the laboratory, do not always develop as well as in the human embryo. This makes the results all the more important for the research team led by Professor Dr. Heiner Niemann, honorary professor at the Hannover Medical School (MHH), and until August 2018 head of the Institute for Farm Animal Genetics in Mariensee at the Friedrich Loeffler Institute for Animal Health.
For the first time, an international research group consisting of scientists from Germany, Great Britain, and China has succeeded in producing a particularly versatile type of stem cells in pigs and humans and to reproduce them under laboratory conditions. Using a special nutrient solution, the scientists were able to reproduce a particularly viable cell type: embryonic stem cell lineages with enhanced potential.
“The EPSC (Expanded Potential Stem Cells ) we produce is something very special because it has a greater development potential than normal embryonic stem cells,” said Professor Niemann. “They care not not only able to develop into all cell types, but also form extraembryonic tissue that surrounds the embryo and later forms important parts of the placenta – the trophoblasts.
Normally, only fertilized oocytes, (before the actual cell division and individual blastomeres at the very beginning of development up until the four- to eight-cell stage), would have this so-called pluripotency the researchers emphasize. Previously, such pluripotent EPSC could only be produced from mice. The researchers have now succeeded in reprogramming pig cells as well as human embryonic stem cells – or induced pluripotent stem cells – to the early pluripotent stage. “Now that the biochemical signaling pathways have been determined more precisely, it is also possible to obtain EPSC from humans and pigs and then reproduce them under established cell culture conditions,” said Niemann.

Deliberate creation of developmental states
“Reprogramming cells into the original stage of a fertilized egg now offers the possibility of producing cell cultures with embryonic properties without actually having to use embryos for research purposes,” explains Niemann. Over the course of the study, the research team investigated the different developmental stages of stem cells and the underlying molecular mechanisms. The researchers succeeded in biochemically inhibiting certain developmental pathways of embryonic stem cells in order to specifically establish desired developmental states, examine disease progressions, or examine developmental disorders.
Niemann believes that pig cells in particular offer ideal conditions for this. “The pig is an increasingly important model for biomedical research,” he emphasizes. Pigs are very similar to humans, their organs are about the same size and research on pig cells is better established than on human cells. In addition, pig cells are easier to genetically modify because the protocols are well established. “In humans, for example, we have to consider tissue incompatibilities,” explains Professor Niemann. “It may be possible to produce universally available and compatible tissue from pig cells that can be stored in a cell bank.” This would also open up new possibilities for the replacement of solid pig organs, the so-called xenotransplantation.
It will take some time before this will be possible because the results have to be confirmed by other research groups. “We are making our work available to the scientific community so that it can be reviewed to prove that the data is solid.” And interest is huge. “At the moment we are being overwhelmed with inquiries,” says Niemann. Cells would constantly have to be packaged and sent around the world so that others could follow the experiments. At the same time, Niemann is already working on the next research proposal. He estimates that the first tissue banks based on the EPSC lineages could be established in three to five years.
The results of the study were published in the journal “Nature Cell Biology“.