TU Delft scientists provide insight into the limitations of super-resolution microscopy and offer a new calculation method to determine maximum resolution. The major precision improvements previously made by fellow-researchers is nuanced by their publication. The technology is important for studying processes in the living cell, discovering the origin of diseases and developing new medicines. In addition, their publication nuances major precision improvements previously claimed by fellow researchers. Their findings were published in Biophysical Journal, writes the TU Delft in a press release.
Delft research
In 2019, researchers from Delft had already given the field of super-resolution microscopy a considerable boost by improving the precision of the technique about twofold (see here). Now they have published a scientific article that points out the fundamental limitations of super-resolution microscopy. ‘We also provide a method for other researchers to help them make more informed choices,’ says Delft PhD student and first author of the publication, Dylan Kalisvaart.
The researchers, led by Carlas Smith, lay new foundations for the super-resolution method called Iterative Single-Molecule Localization Microscopy. They use illumination patterns to zoom in on individual molecules. To do so, they use results from previous experiments to place the patterns closer and closer to molecules. This makes it possible to increase the sharpness of the image exactly where the molecules are.
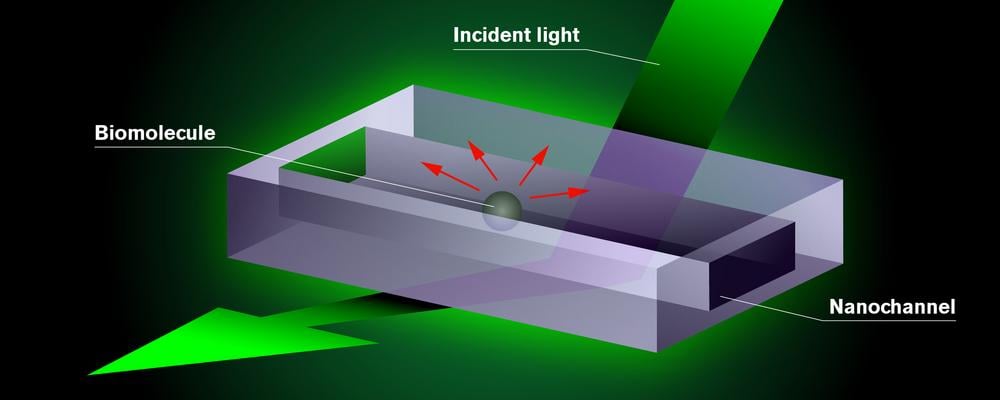
Kalisvaart, researcher at the Delft Center for Systems and Control, explains: ‘We show (with the so-called Van Trees inequality) that resolution improvements can be attributed to prior knowledge obtained from previous experiments. This enables us to demonstrate what the practical settings of a microscope should be, given the circumstances and prior knowledge, in order to achieve the best result.’
Super-resolution microscopy
Super-resolution microscopy is a groundbreaking technology that allows researchers to look inside living cells. The technique uses luminescent proteins that can be found in jellyfish, for example. In 2008, three top researchers were awarded the Nobel Prize in Chemistry for the discovery and development of this luminescent protein, called GFP (Green Fluorescent Protein). Researchers can attach these fluorescent proteins to molecules using gene editing. When you shine a laser on these proteins, they emit a small amount of light.
Single Molecule Localization Microscopy (SMLM) ensure that molecules are randomly switched on or off. Sensitive sensors make a video of these light signals, after which researchers analyse the data obtained. This enables them to determine the location of the molecules very precisely and to make a reconstruction of the cell structure. With an ordinary optical microscope, you can make images at a scale of about half a micron. Super-resolution microscopy increases this ability by a factor of ten.
Selected for you!
Innovation Origins is the European platform for innovation news. In addition to the many reports from our own editors in 15 European countries, we select the most important press releases from reliable sources. This way you can stay up to date on what is happening in the world of innovation. Are you or do you know an organization that should not be missing from our list of selected sources? Then report to our editorial team.