Finally a breakthrough after a little over 40 years in research into alpha-A crystallin – a protective protein in the eye lens. Researchers at the Technical University of Munich (TUM) have for the first time succeeded in determining the structure of the protein and thereby discovered one other important function.
These findings help to find new preventative and therapeutic strategies to treat cataracts.
The human eye lens is made of a highly concentrated protein solution which gives it its considerable refractive power. This determines to what extent light rays entering the eye are refracted. A process which is indispensable for keen vision. As it is only when light is deflected by the cornea and the eye lens, that light rays are bundled and hit the retina where vision is at its sharpest.
Crucial for the function of the eye lens
These proteins are created when an embryo is in development. And the proteins have to continue to function for the rest of a person’s life as the eye lens has no built-in mechanism for building and breaking down proteins. If the lens proteins become damaged, presbyopia and cataracts will occur – i.e., a clouding of the eye lens.
These are protective proteins that prevent proteins from clumping together over time. The two protective proteins alpha-A crystallin and alpha-B crystallin make up around 30% of the proteins in the human eye and are crucial for the function of the eye lens.
Decoding the structure of alpha-A crystallin
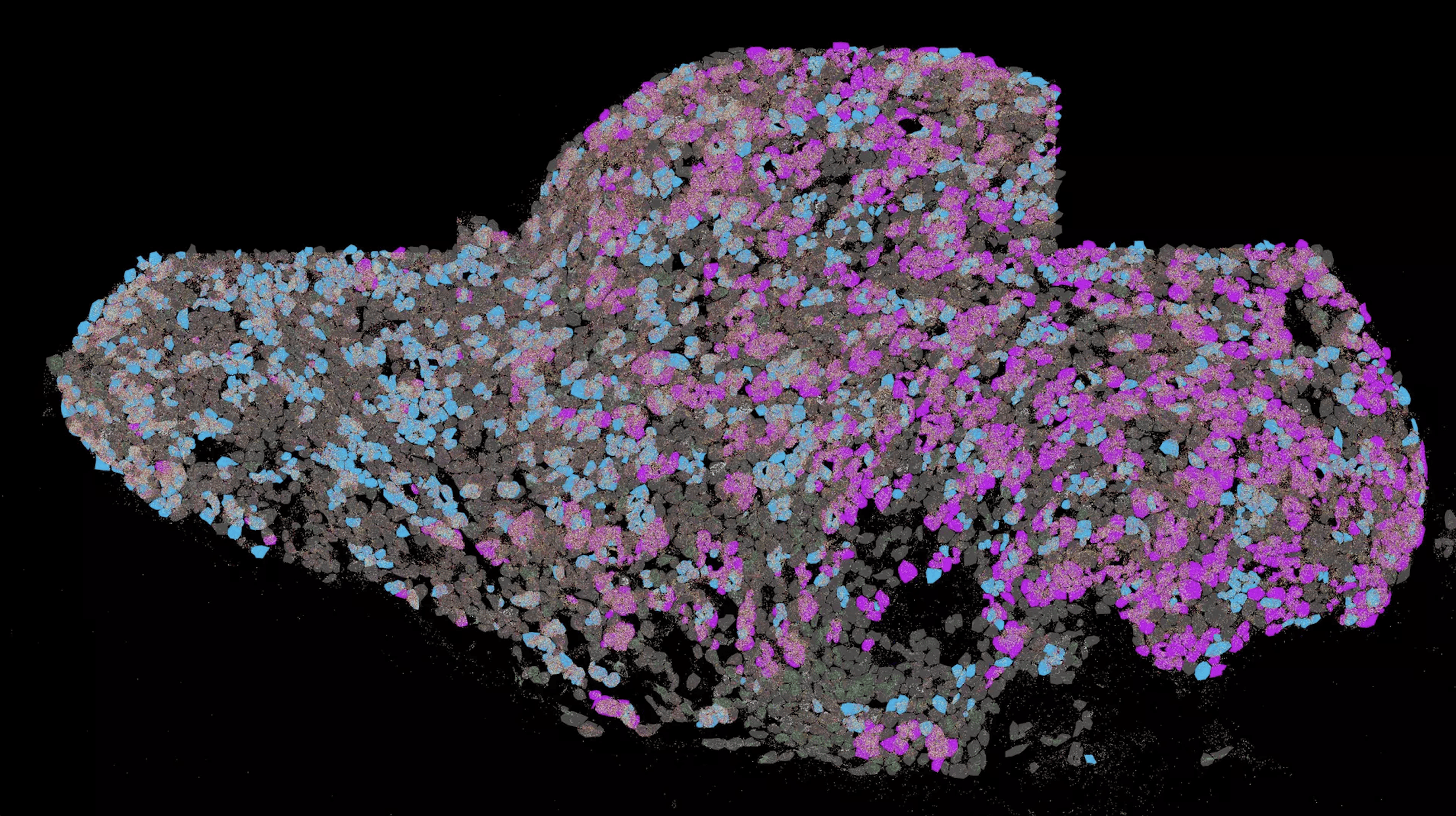
Researchers have been working on decoding the structure of alpha-A crystallin for forty years. A research team led by Sevil Weinkauf, Professor of Electron Microscopy and Johannes Buchner, Professor of Biotechnology at the Technical University of Munich (TUM) have finally made a breakthrough. They developed a new strategy for data analysis and combined cryo-electron microscopy, mass spectrometry, Nuclear Magnetic Resonance spectroscopy and molecular modelling. They found that the structure of the alpha-A crystalline is present in solutions in several structures with 12, 16 or 20 subunits.

Photo: Human alpha-A crystallin is present in structures of 12, 16 and 20 subsets respectively (from left to right). Two subunits each ( center, dark and light grey) form a dimer. A dimer is a molecule or a molecule compound consisting of the monomers, two often identical subunits.
Role of protective proteins
Protective proteins are able to help other proteins under stress to retain their shape. Stress can be caused by high temperatures, for example. Alpha-A crystallin and alpha-B crystallin also have this function. Moreover, human alpha-A crystallin consists of two cysteine residues whose sulfur atoms are capable of forming disulfide bridges. Cysteine is an amino acid and one of the 20 building blocks of protein.
Biochemical research has shown that this type of bridging has a significant effect on various properties of the protein molecule.
Protecting other proteins from oxidation
The findings of the researchers suggest that alpha-A crystallin plays an active role in protecting other proteins from oxidation.
It was found that oxidized alpha-A crystallin can even transfer the disulfide bridge present to other proteins and thereby influence the redox state of other lens proteins. Redox is a combination of reduction and oxidation. This function also explains why approximately half of the alpha-A crystallins already have this type of disulfide bridge in embryos.
About 35% of all cases of blindness are attributed to cataracts. The molecular insight into the functions of the eye lens proteins helps to develop new preventative and therapeutic strategies. Recognizing that alpha-A crystallin also plays an important role in oxidation resistance is fundamental for future research projects.
Also of interest:
Digital imaging module for improving diagnostics in ophthalmology
New production process for improved protein therapeutics