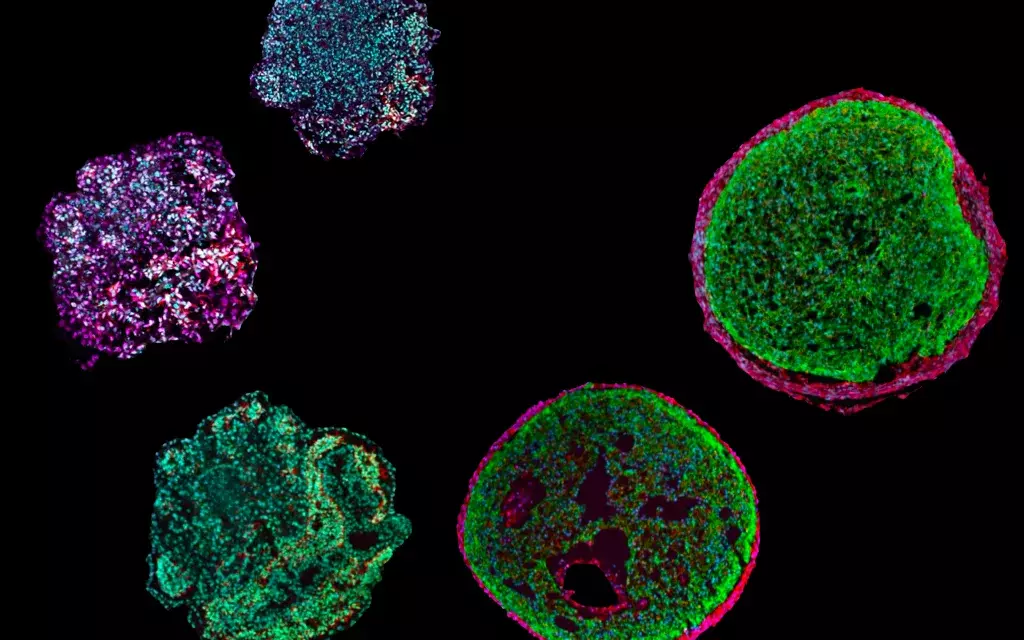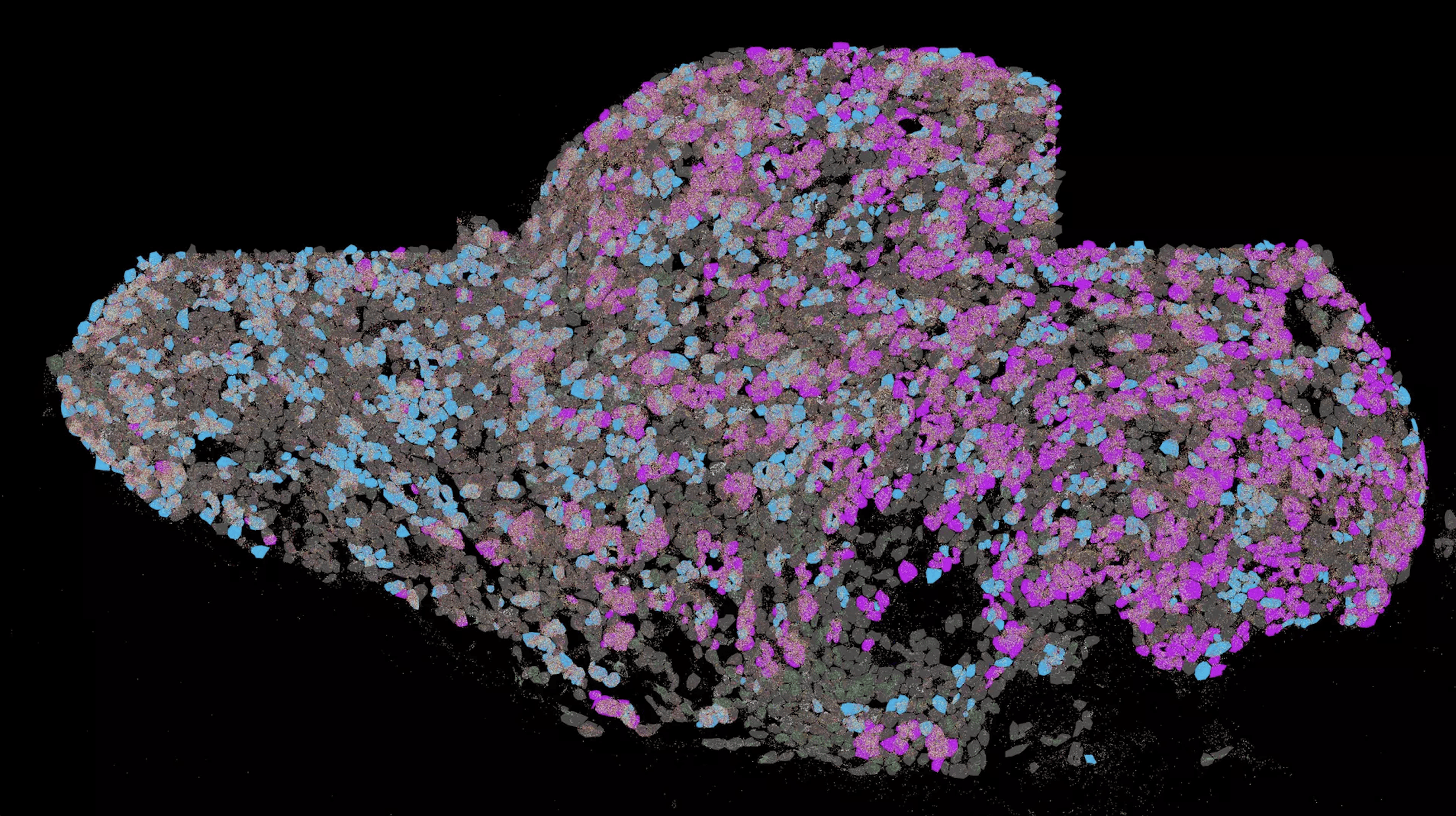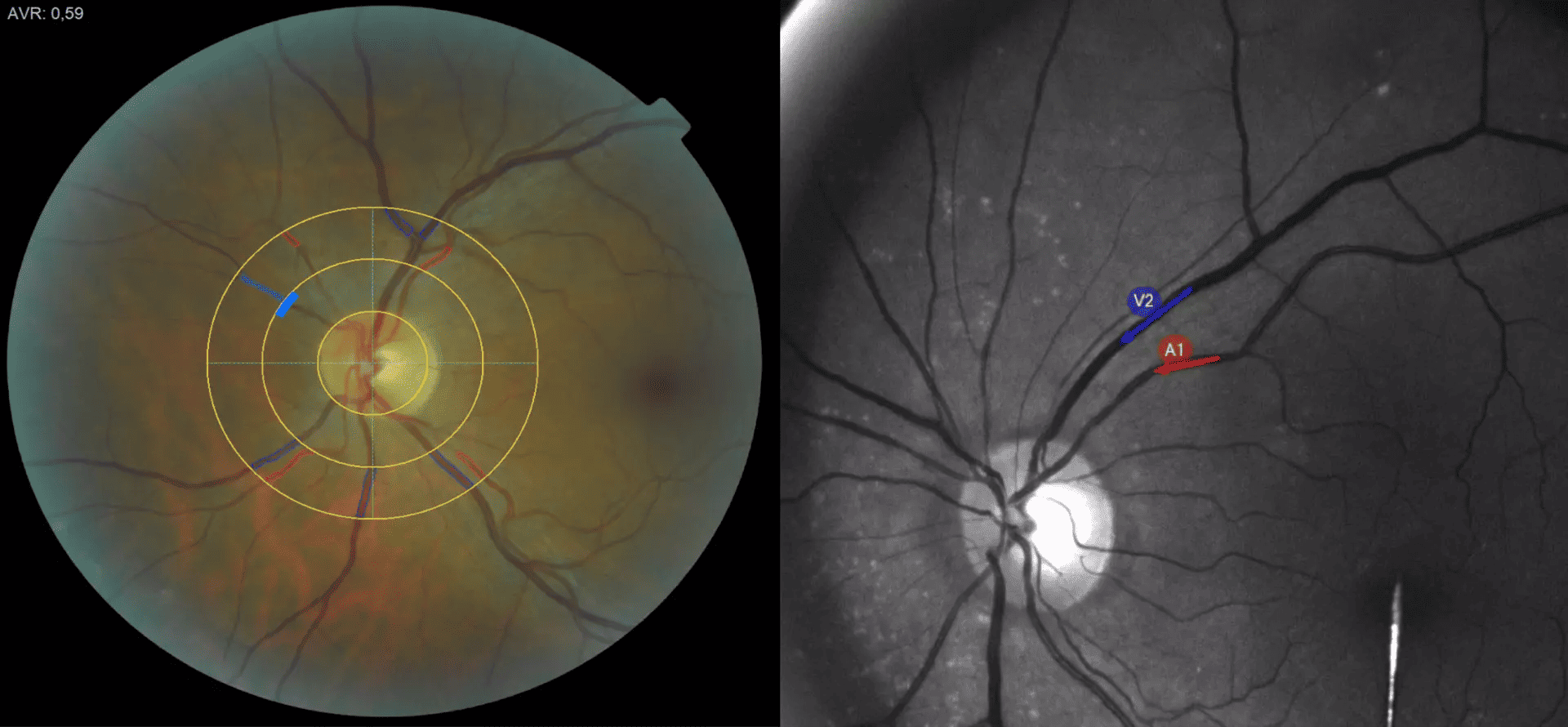
In general, the human heart beats around 60 to 80 times per minute, but in about one million people in Germany – more than 33 million worldwide – the heart rate can suddenly increase to up to 150 beats. These people suffer from atrial fibrillation, in Germany the most common type of cardiac arrhythmia requiring treatment. While atrial fibrillation in itself is generally not life-threatening, its consequences such as heart failure, heart attack, blood clots, and strokes can result in serious consequences or even death.
A typical treatment for fibrillation is catheter-assisted sclerotherapy of the electrophysiological excitation centers inside the heart muscle tissue, known as cardiac ablation. Nevertheless, it often happens that patients develop an atypical atrial flutter after treatment. Unlike in atrial fibrillation, with atrial flutter the electrophysiological excitation of the heart is coordinated. In this process, unusually fast electrical excitation patterns cause the atria to contract rapidly. However, the effects are the same as with atrial fibrillation: Cardiac tachycardia, shortness of breath and general weakness. The risk of stroke is also increased.
Realistic computer models of the heart
Up until now, there has been no reliable way to investigate how high the risk is for patients when it comes to developing atypical atrial flutter. Thanks to digital simulations of human organs, however, it is now possible to explore the development of diseases and tailor therapies to patients. Researchers at the Karlsruhe Institute of Technology (KIT), the Medical Clinic IV of the Karlsruhe Municipal Hospital, the Medical Faculty of the University of Freiburg, and the University Heart Center Freiburg-Bad Krozingen have developed a method that is able to assess the risk of atrial flutter on an individual basis.

They are developing realistic computer models of the heart across several levels: from the cardiac ion channel, to the cells and tissue and the entire organ. They simulate basic physiological and pathological processes and develop personalized models in order to assess the risk of cardiac arrhythmias, such as atrial flutter, along with the effect of individual therapies. In the journal Frontiers in Physiology, the scientists report that personalized computer models can “identify all paths along which atypical, circular electrical excitation can occur.”
“Our models include anatomical, electrophysiological, and pharmacological criteria,” explains Dr. Axel Loewe, head of the cardiac modeling workgroup at the Institute of Biomedical Engineering at KIT. The effect of therapies such as catheter ablation or medications are also able to be individually assessed in advance. “Computer models offer a perfectly controllable environment for experiments. This way, individual variables can be simulated and their consequences calculated for the overall system.” According to the researchers, the models would complement classical methods such as cell and animal experiments and make it possible to be able to test new therapies without risk for humans.
Interdisciplinary cooperation
In his dissertation, Loewe has already simulated the causes of atrial fibrillation with a computer. He now heads the KIT cardiac modeling workgroup which is developing new computer models of the heart. Using these models, scientists are able to simulate how electrophysilogical excitation develops and spreads through the atria and the entire heart. In a healthy beating heart, this excitation ceases, but it persists with certain types of cardiac arrhythmias.
In addition, scientists are working with personalized models in order to be able to determine on an individual basis the risk of disease and the effect of treatment. Using imaging techniques such as magnetic resonance imaging (MRI), they record the individual anatomy of a patient, such as the size and shape of the atria. An electrocardiogram (ECG) records the electrical activity of the heart. The scientists are working closely with the Bioelectrical Signals workgroup at the KIT Institute of Biomedical Engineering headed by Professor Olaf Dössel. “The work, which ranges between engineering sciences, computer science, natural sciences, and medicine, paves the way for tailor-made therapies,” the researchers explain.
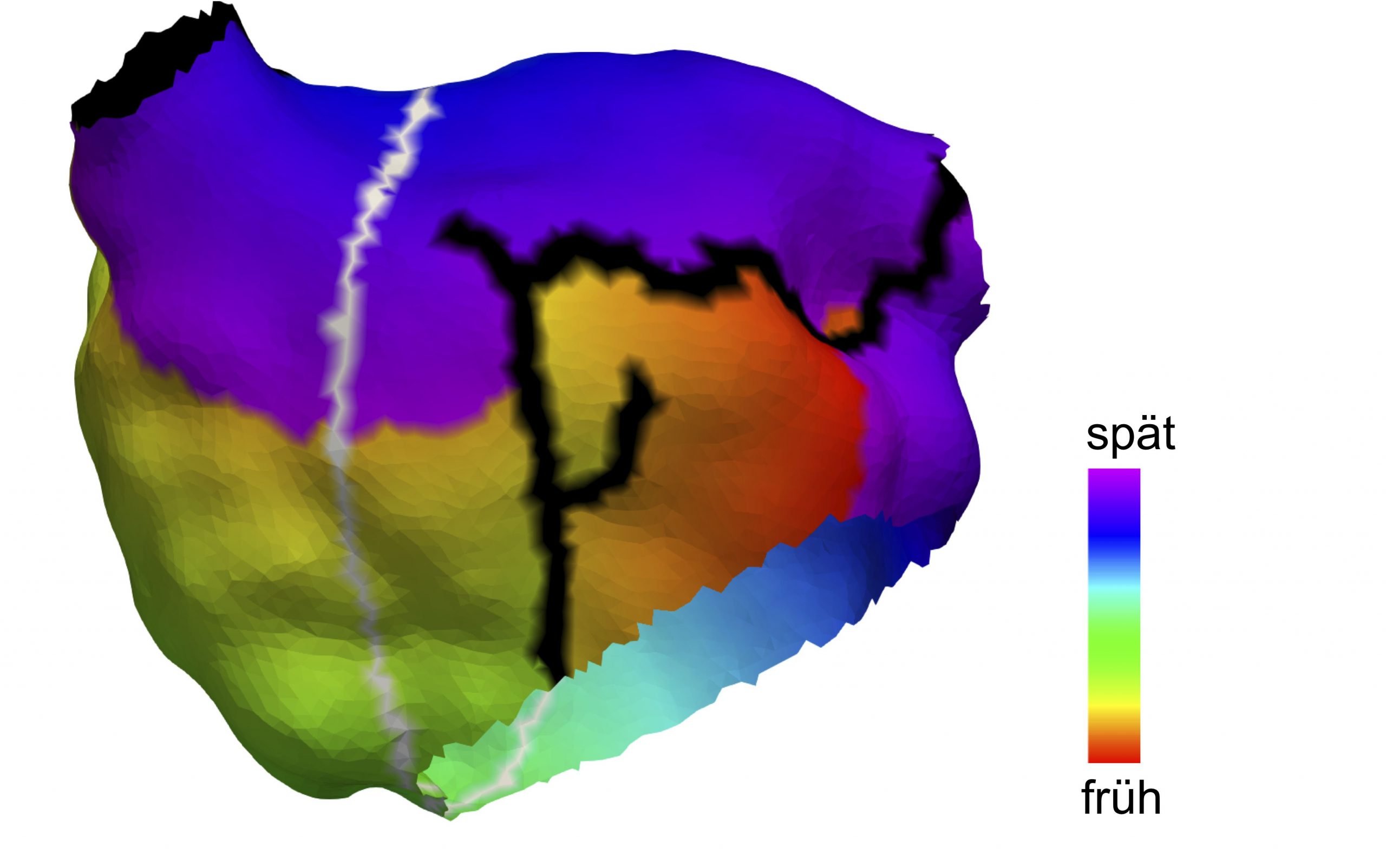
Cover picture: Anatomical model of the left atrium of a 70-year-old female patient. (Illustration: Axel Loewe, KIT)